13.4: Effects Of Temperature And Pressure On Solubility
Maybe your like
Effect of Temperature on the Solubility of Solids
Figure \(\PageIndex{1}\) shows plots of the solubilities of several organic and inorganic compounds in water as a function of temperature. Although the solubility of a solid generally increases with increasing temperature, there is no simple relationship between the structure of a substance and the temperature dependence of its solubility. Many compounds (such as glucose and \(\ce{CH_3CO_2Na}\)) exhibit a dramatic increase in solubility with increasing temperature. Others (such as \(\ce{NaCl}\) and \(\ce{K_2SO_4}\)) exhibit little variation, and still others (such as \(\ce{Li_2SO_4}\)) become less soluble with increasing temperature.
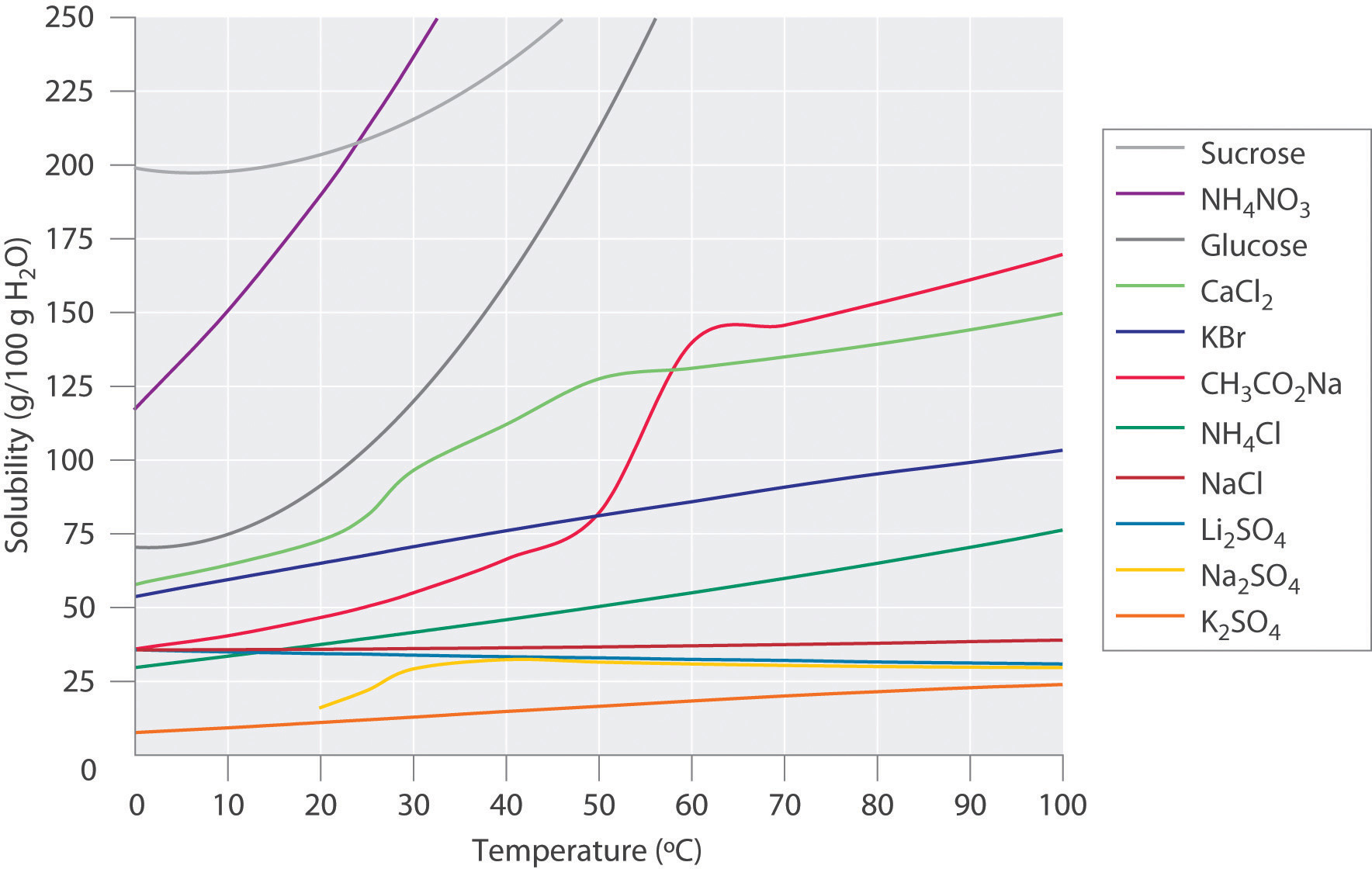
Notice in particular the curves for \(\ce{NH4NO3}\) and \(\ce{CaCl2}\). The dissolution of ammonium nitrate in water is endothermic (\(ΔH_{soln} = +25.7\; kJ/mol\)), whereas the dissolution of calcium chloride is exothermic (\(ΔH_{soln} = −68.2 \;kJ/mol\)), yet Figure \(\PageIndex{1}\) shows that the solubility of both compounds increases sharply with increasing temperature. In fact, the magnitudes of the changes in both enthalpy and entropy for dissolution are temperature dependent. Because the solubility of a compound is ultimately determined by relatively small differences between large numbers, there is generally no good way to predict how the solubility will vary with temperature.
The variation of solubility with temperature has been measured for a wide range of compounds, and the results are published in many standard reference books. Chemists are often able to use this information to separate the components of a mixture by fractional crystallization, the separation of compounds on the basis of their solubilities in a given solvent. For example, if we have a mixture of 150 g of sodium acetate (\(\ce{CH_3CO_2Na}\)) and 50 g of \(\ce{KBr}\), we can separate the two compounds by dissolving the mixture in 100 g of water at 80°C and then cooling the solution slowly to 0°C. According to the temperature curves in Figure \(\PageIndex{1}\), both compounds dissolve in water at 80°C, and all 50 g of \(\ce{KBr}\) remains in solution at 0°C. Only about 36 g of \(\ce{CH3CO2Na}\) are soluble in 100 g of water at 0°C, however, so approximately 114 g (150 g − 36 g) of \(\ce{CH_3CO_2Na}\) crystallizes out on cooling. The crystals can then be separated by filtration. Thus fractional crystallization allows us to recover about 75% of the original \(\ce{CH_3CO_2Na}\) in essentially pure form in only one step.
Fractional crystallization is a common technique for purifying compounds as diverse as those shown in Figure \(\PageIndex{1}\) and from antibiotics to enzymes. For the technique to work properly, the compound of interest must be more soluble at high temperature than at low temperature, so that lowering the temperature causes it to crystallize out of solution. In addition, the impurities must be more soluble than the compound of interest (as was \(\ce{KBr}\) in this example) and preferably present in relatively small amounts.
Tag » When Temperature Decreases What Happens To Pressure
-
Gas Laws
-
How Does A Decrease In Temperature Affect The Pressure Of A ...
-
What Happens To The Gas Pressure When The Temperature Decreases?
-
PRESSURE AND TEMPERATURE RELATIONSHIP
-
What Happens When The Pressure & Temperature Of A Fixed ...
-
How Does Temperature Affect The Pressure Of A Gas? - Pico Technology
-
Ideal Gas Law
-
Temperature And Pressure Effects
-
Vapor Pressure
-
What Happens To Pressure If Temperature Increases? - Byju's
-
9.2 Relating Pressure, Volume, Amount, And Temperature: The Ideal ...
-
The Effect Of Changing Conditions - Chemistry LibreTexts
-
Effects Of Temperature And Pressure On State Science Games
-
It Is A Rule That As We Go Up,the Air Pressure Decreases.And ... - Byju's