4.5: Defining Isotopes - Chemistry LibreTexts
Maybe your like
Isotopes
How many neutrons are in atoms of a particular element? At first it was thought that the number of neutrons in a nucleus was also characteristic of an element. However, it was found that atoms of the same element can have different numbers of neutrons. Atoms of the same element (i.e., same atomic number, Z) that have different numbers of neutrons are called isotopes. For example, 99% of the carbon atoms on Earth have 6 neutrons and 6 protons in their nuclei; about 1% of the carbon atoms have 7 neutrons in their nuclei. Naturally occurring carbon on Earth, therefore, is actually a mixture of isotopes, albeit a mixture that is 99% carbon with 6 neutrons in each nucleus.
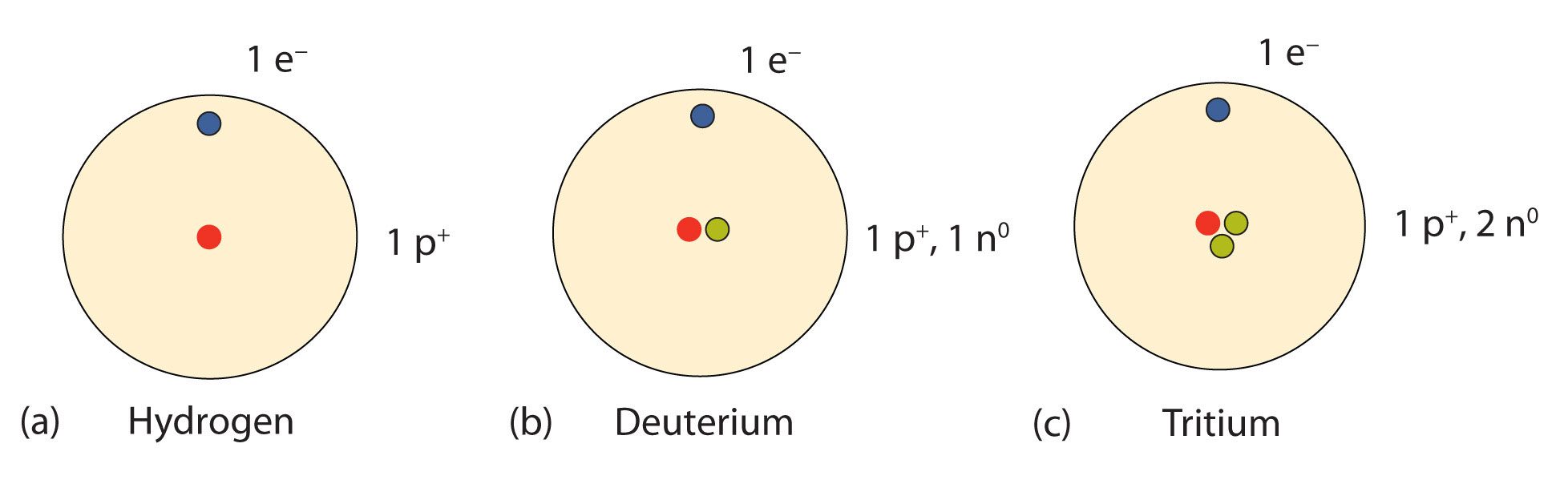
An important series of isotopes is found with hydrogen atoms. Most hydrogen atoms have a nucleus with only a single proton. About 1 in 10,000 hydrogen nuclei, however, also has a neutron; this particular isotope is called deuterium. An extremely rare hydrogen isotope, tritium, has 1 proton and 2 neutrons in its nucleus. Figure \(\PageIndex{1}\) compares the three isotopes of hydrogen.
The discovery of isotopes required a minor change in Dalton’s atomic theory. Dalton thought that all atoms of the same element were exactly the same.
Most elements exist as mixtures of isotopes. In fact, there are currently over 3,500 isotopes known for all the elements. When scientists discuss individual isotopes, they need an efficient way to specify the number of neutrons in any particular nucleus. The mass number (A) of an atom is the sum of the numbers of protons and neutrons in the nucleus. Given the mass number for a nucleus (and knowing the atomic number of that particular atom), you can determine the number of neutrons by subtracting the atomic number from the mass number.
A simple way of indicating the mass number of a particular isotope is to list it as a superscript on the left side of an element’s symbol. Atomic numbers are often listed as a subscript on the left side of an element’s symbol. Thus, we might see
\[\mathrm{^{mass\: number\xrightarrow{\hspace{45px}} 56}_{atomic\: number \xrightarrow{\hspace{35px}} 26}Fe} \label{Eq1} \]
which indicates a particular isotope of iron. The 26 is the atomic number (which is the same for all iron atoms), while the 56 is the mass number of the isotope. To determine the number of neutrons in this isotope, we subtract 26 from 56: 56 − 26 = 30, so there are 30 neutrons in this atom.
Example \(\PageIndex{3}\)
How many protons and neutrons are in each atom?
- \(\mathrm{^{35}_{17}Cl}\)
- \(\mathrm{^{127}_{53}I}\)
In \(\mathrm{^{35}_{17}Cl}\) there are 17 protons, and 35 − 17 = 18 neutrons in each nucleus.
Answer bIn \(\mathrm{^{127}_{53}I}\) there are 53 protons, and 127 − 53 = 74 neutrons in each nucleus.
Exercise \(\PageIndex{3}\)
How many protons and neutrons are in each atom?
- \(\mathrm{^{197}_{79}Au}\)
- \(\mathrm{^{23}_{11}Na}\)
In \(\mathrm{^{197}_{79}Au}\) there are 79 protons, and 197 − 79 = 118 neutrons in each nucleus.
Answer bIn \(\mathrm{^{23}_{11}Na}\) there are 11 protons, and 23 − 11 = 12 neutrons in each nucleus.
It is not absolutely necessary to indicate the atomic number as a subscript because each element has its own unique atomic number. Many isotopes are indicated with a superscript only, such as 13C or 235U. You may also see isotopes represented in print as, for example, carbon-13 or uranium-235.
Tag » What Is A Neutral Atom
-
Atoms Vs. Ions
-
Definition And Examples Of Neutral Atom Along With FAQs - Byju's
-
Why Do Atoms Always Contain The Same Number Of Electrons And ...
-
What Is A Neutral Atom? - Chemistry Stack Exchange
-
What Is A Neutral Atom ? Definition And Examples - Basic Mathematics
-
Neutral Atom - An Overview | ScienceDirect Topics
-
What Is A Neutral Atom? - Socratic
-
What Is A Neutral Atom? | General Knowledge Questions - Toppr
-
What Does 'neutral Atom' Mean? - Quora
-
Why Are Atoms Neutral? | GCSE Chemistry (9-1)
-
What Is A Neutral Atom? - YouTube
-
Neutral Atom - YouTube
-
Energetic Neutral Atom - Wikipedia
-
Neutral Atom In A Sentence - Cambridge Dictionary