9.6: Single Covalent Bonds - Chemistry LibreTexts
Maybe your like
Single Covalent Bonds
A covalent bond forms when two orbitals with one electron each overlap one another. For the hydrogen molecule, this can be shown as:
Upon formation of the \(\ce{H_2}\) molecule, the shared electrons must have opposite spin, so they are shown with opposite spin in the atomic \(1s\) orbital.
The halogens also form single covalent bonds in their diatomic molecules. An atom of any halogen, such as fluorine, has seven valence electrons. Its unpaired electron is located in the \(2p\) orbital.
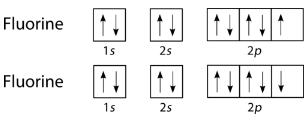
The single electrons in the third \(2p\) orbital combine to form the covalent bond:
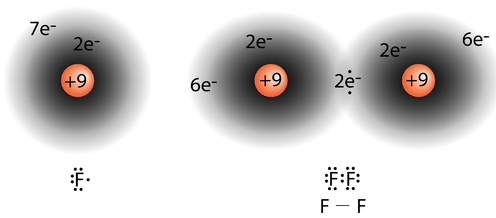
The diatomic fluorine molecule \(\left( \ce{F_2} \right)\) contains a single shared pair of electrons. Each \(\ce{F}\) atom also has three pair of electrons that are not shared with the other atom. A lone pair is a pair of electrons in a Lewis electron-dot structure that is not shared between atoms. The oxygen atom in the water molecule shown below has two lone pair sets of electrons. Each \(\ce{F}\) atom has three lone pairs. Combined with the two electrons in the covalent bond, each \(\ce{F}\) atom follows the octet rule.
Example \(\PageIndex{1}\)
Draw the Lewis electron dot structure for water.
Solution
Step 1: List the known quantities and plan the problem.
Known
- Molecular formula of water \(= \ce{H_2O}\)
- \(1 \: \ce{O}\) atom \(= 6\) valence electrons
- \(2 \: \ce{H}\) atoms \(=2 \times 1 = 2\) valence electrons
- Total number of valence electrons \(=8\)
Use the periodic table to determine the number of valence electrons for each atom and the total number of valence electrons. Arrange the atoms and distribute the electrons so that each atom follows the octet rule. The oxygen atom will have 8 electrons, while the hydrogen atoms will each have 2.
Step 2: Solve.
Electron dot diagrams for each atom are:
Each hydrogen atom with its single electron will form a covalent bond with the oxygen atom where it has a single electron. The resulting Lewis electron dot structure is:
Step 3: Think about your result.
The oxygen atom follows the octet rule with two pairs of bonding electrons and two lone pairs. Each hydrogen atom follows the octet rule with one bonding pair of electrons.
Tag » How Many Electrons Are Shared In A Single Covalent Bond
-
Covalent Bond - An Overview | ScienceDirect Topics
-
How Many Electrons Are Shared In A Single Bond And A Double Bond?
-
How Many Electrons Are Shared In A Single Covalent Bond? - Toppr
-
How Many Shared Electrons Result In A Single Covalent Bond? - Byju's
-
Single And Multiple Covalent Bonds (article) - Khan Academy
-
Single Covalent Bonds Study Guide | Inspirit
-
4.6: Covalent Bonds- Shared Electron Pairs - Chemistry LibreTexts
-
How Many Electrons Are Shared In A Single Bond Between ... - Nagwa
-
Covalent-bonding-and-molecules - Miss Chen
-
6.3 Covalent Bonding | Chemical Bonding - Siyavula
-
Covalent Bonds - Life Sciences Cyberbridge
-
Single Bond - Wikipedia
-
Covalent Compounds - University Of Hawaii At Manoa
-
9.3 Covalent Bonds