A Subunit Vaccine Candidate Based On The Spike Protein Of ... - PubMed
Maybe your like
Abstract
Of the numerous animal species affected by the SARS-CoV-2 virus, cats are one of the most susceptible, and cat-to-cat transmission has been described. Although cat-to-human infection has not, as yet, been demonstrated, preventive measures should be taken in order to avoid both viral infection in cats and transmission among them. In this respect, the application of an effective vaccine to at-risk populations would be a useful tool for controlling the disease in this species. Here, we test a new vaccine prototype based on the Spike protein of the virus in order to prevent infection and infectious virus shedding in cats. The vaccine employed in experimentation, and which is easily produced, triggered a strong neutralizing antibody response in vaccinated animals. In contrast to that which occurred with control animals, no infectious virus was detected in the oropharyngeal or rectal swabs of vaccinated cats submitted to a SARS-CoV-2 challenge. These results are of great interest as regards future considerations related to implementing vaccination programs in pets. The value of cats as vaccination trial models is also described herein.
Keywords: Cats; SARS-CoV-2; Spike protein; Subunit vaccine.
Copyright © 2022 The Authors. Published by Elsevier Ltd.. All rights reserved.
PubMed Disclaimer
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Fig. 1
Outline of the procedure carried…
Fig. 1
Outline of the procedure carried out during the experiment on animals VAC1, VAC2,…
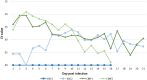
Fig. 2
Average value of Ct in…
Fig. 2
Average value of Ct in oropharyngeal swabs of animals VAC1 (euthanized on 16…

Fig. 3
Percentage of inhibition in VAC1…
Fig. 3
Percentage of inhibition in VAC1 and VAC2 from 20 DPV (day post-vaccination; one…
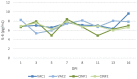
Fig. 4
Interleukin 6 (IL-6) levels in…
Fig. 4
Interleukin 6 (IL-6) levels in serum in vaccinated cats (VAC1 and VAC2) and…

Fig. 5
Interferon-gamma (IFN- γ) levels in…
Fig. 5
Interferon-gamma (IFN- γ) levels in serum in vaccinated cats (VAC1 and VAC2) and…
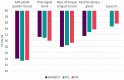
Fig. 6
Viral loads based on Ct…
Fig. 6
Viral loads based on Ct values in tissues from animal VAC2 euthanized on…

Fig. 7
Viral loads based on Ct…
Fig. 7
Viral loads based on Ct values in tissues from animal CINF1 euthanized on…

Fig. 8
Viral loads based on Ct…
Fig. 8
Viral loads based on Ct values in tissues from animal CINF2 euthanized on…

Fig. 9
Pulmonary lesions found in vaccinated…
Fig. 9
Pulmonary lesions found in vaccinated and non-vaccinated cats after SARS-CoV-2 infection. The lungs…
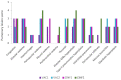
Fig. 10
Pulmonary histopathological findings and their…
Fig. 10
Pulmonary histopathological findings and their corresponding semi-quantitative grades of evaluation in VAC1, VAC2,…
References
-
- Baden L.R., El Sahly H.M., Essink B., Kotloff K., Frey S., Novak R., Diemert D., Spector S.A., Rouphael N., Creech C.B., McGettigan J., Khetan S., Segall N., Solis J., Brosz A., Fierro C., Schwartz H., Neuzil K., Corey L., Gilbert P., Janes H., Follmann D., Marovich M., Mascola J., Polakowski L., Ledgerwood J., Graham B.S., Bennett H., Pajon R., Knightly C., Leav B., Deng W., Zhou H., Han S., Ivarsson M., Miller J., Zaks T. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021;384:403–416. - PMC - PubMed
-
- Barroso-Arévalo S., Barneto A., Ramos Á.M., Rivera B., Sánchez R., Sánchez-Morales L., Pérez-Sancho M., Buendía A., Ferreras E., Ortiz-Menéndez J.C., Moreno I., Serres C., Vela C., Risalde M.Á., Domínguez L., Sánchez-Vizcaíno J.M. Large-scale study on virological and serological prevalence of SARS-CoV-2 in cats and dogs in Spain. Transbound. Emerg. Dis. 2021;1(10):1–16. doi: 10.1111/tbed.14366. - DOI - PMC - PubMed
-
- Barroso-Arévalo S., Sánchez-Morales L., Barasona J.A., Rivera B., Sánchez R., Risalde M.A., Agulló-Ros I., Sánchez-Vizcaíno J.M. Evaluation of the clinical evolution and transmission of SARS-CoV-2 infection in cats by simulating natural routes of infection. Vet. Res. Commun. 2022;3(1–16) doi: 10.1007/s11259-022-09908-5. - DOI - PMC - PubMed
-
- Bosco-Lauth A.M., Hartwig A.E., Porter S.M., Gordy P.W., Nehring M., Byas A.D., VandeWoude S., Ragan I.K., Maison R.M., Bowen R.A. Experimental infection of domestic dogs and cats with SARS-CoV-2: pathogenesis, transmission, and response to reexposure in cats. Proc. Natl. Acad. Sci. 2020;117:26382. - PMC - PubMed
-
- Bukreyev A., Lamirande E.W., Buchholz U.J., Vogel L.N., Elkins W.R., St Claire M., Murphy B.R., Subbarao K., Collins P.L. Mucosal immunisation of African green monkeys (Cercopithecus aethiops) with an attenuated parainfluenza virus expressing the SARS coronavirus spike protein for the prevention of SARS. Lancet. 2004;363:2122–2127. - PMC - PubMed
MeSH terms
- Animals Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Antibodies, Neutralizing Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- COVID-19* / prevention & control Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- COVID-19* / veterinary Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Cat Diseases* / prevention & control Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Cats Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- SARS-CoV-2 Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Spike Glycoprotein, Coronavirus Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Vaccines, Subunit Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Virus Shedding Actions
- Search in PubMed
- Search in MeSH
- Add to Search
Substances
- Antibodies, Neutralizing Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Spike Glycoprotein, Coronavirus Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Vaccines, Subunit Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- spike protein, SARS-CoV-2 Actions
- Search in PubMed
- Search in MeSH
- Add to Search
LinkOut - more resources
Full Text Sources
- Andalusian Health Repository - access to free full text
- Elsevier Science
- Europe PubMed Central
- PubMed Central
Medical
- MedlinePlus Health Information
Miscellaneous
- NCI CPTAC Assay Portal
Tag » What Is Spike Protein Shedding
-
Do Vaccinated People Shed Spike Proteins Or The Virus Could Be ...
-
COVID-19 Vaccines, Irregular Periods And Spike Protein Shedding
-
Adverse Effects Of COVID-19 MRNA Vaccines: The Spike Hypothesis
-
Viral Shedding And COVID-19 — What Can And Can't Happen
-
A Subunit Vaccine Candidate Based On The Spike Protein Of SARS ...
-
Respiratory Viral Shedding In Healthcare Workers Reinfected ... - CDC
-
Fact Check-Those Vaccinated For COVID-19 Are Not Shedding Toxic ...
-
Can COVID Vaccines Shed Spike Proteins – And Is That Bad?
-
People Vaccinated Against COVID-19 Cannot 'shed' Spike Proteins To ...
-
Pregnancies, Periods And COVID-19 Vaccines: What You Need To Know
-
[PDF] Worse Than The Disease? Reviewing Some Possible Unintended ...
-
MRNA Technology Pioneer Says Covid-19 Vaccinated People Can ...
-
No Scientific Basis For Vaccine 'shedding' Claims | FactCheck
-
Urinary Shedding Of Spike Protein In COVID-19 Patients