Comparative Analysis Of TCR And CAR Signaling Informs ... - PubMed
Maybe your like
Abstract
Chimeric antigen receptor (CAR)-modified T cell therapy is effective in treating lymphomas, leukemias, and multiple myeloma in which the tumor cells express high amounts of target antigen. However, achieving durable remission for these hematological malignancies and extending CAR T cell therapy to patients with solid tumors will require receptors that can recognize and eliminate tumor cells with a low density of target antigen. Although CARs were designed to mimic T cell receptor (TCR) signaling, TCRs are at least 100-fold more sensitive to antigen. To design a CAR with improved antigen sensitivity, we directly compared TCR and CAR signaling in primary human T cells. Global phosphoproteomic analysis revealed that key T cell signaling proteins-such as CD3δ, CD3ε, and CD3γ, which comprise a portion of the T cell co-receptor, as well as the TCR adaptor protein LAT-were either not phosphorylated or were only weakly phosphorylated by CAR stimulation. Modifying a commonplace 4-1BB/CD3ζ CAR sequence to better engage CD3ε and LAT using embedded CD3ε or GRB2 domains resulted in enhanced T cell activation in vitro in settings of a low density of antigen, and improved efficacy in in vivo models of lymphoma, leukemia, and breast cancer. These CARs represent examples of alterations in receptor design that were guided by in-depth interrogation of T cell signaling.
Copyright © 2021 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
PubMed Disclaimer
Conflict of interest statement
Competing interests: A.I.S. is a shareholder of and scientific consultant for Lyell Immunopharma. S.R.R. is a founder and shareholder of Lyell Immunopharma, and was a founder and scientific advisor of Juno Therapeutics, a Celgene company. R.G. has received consulting income from Juno Therapeutics, Celgene, Illumina, Takeda, Infotech Soft, and Merck, research support from Janssen Pharmaceuticals and Juno Therapeutics, and has an ownership interest in CellSpace Biosciences, Ozette Technologies, and Modulus Therapeutics. A.I.S., A.R., and S.R.R. have filed provisional patents relating to the CAR designs described in this manuscript. The other authors declare that they have no competing interests.
Figures
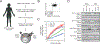
Figure 1.. Bi-specific T cells and magnetic…
Figure 1.. Bi-specific T cells and magnetic beads enable analysis of TCR and CAR signaling…

Figure 2.. CAR stimulation promotes less intense…
Figure 2.. CAR stimulation promotes less intense phosphorylation of CD3 chains and proximal TCR signaling…

Figure 3.. MS-guided CAR designs are expressed…
Figure 3.. MS-guided CAR designs are expressed by primary T cells.
(A) Schematics of CARs…
Figure 4.. Novel CAR designs possess increased…
Figure 4.. Novel CAR designs possess increased antigen sensitivity.
(A) X-Y plot shows the mean…
Figure 5.. BB/ζ CARs containing CD3ζ or…
Figure 5.. BB/ζ CARs containing CD3ζ or GRB2 domains maintain in vivo antitumor function in…
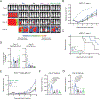
Figure 6.. BB/ζ CARs containing CD3ζ or…
Figure 6.. BB/ζ CARs containing CD3ζ or GRB2 domains enhance in vivo antitumor function in…
References
-
- Turtle CJ, Hanafi L-A, Berger C, Gooley TA, Cherian S, Hudecek M, Sommermeyer D, Melville K, Pender B, Budiarto TM, Robinson E, Steevens NN, Chaney C, Soma L, Chen X, Yeung C, Wood B, Li D, Cao J, Heimfeld S, Jensen MC, Riddell SR, Maloney DG, CD19 CAR-T cells of defined CD4+:CD8+ composition in adult B cell ALL patients, J. Clin. Invest 126, 2123–2138 (2016). - PMC - PubMed
-
- Turtle CJ, Hanafi L-A, Berger C, Hudecek M, Pender B, Robinson E, Hawkins R, Chaney C, Cherian S, Chen X, Soma L, Wood B, Li D, Heimfeld S, Riddell SR, Maloney DG, Immunotherapy of non-Hodgkin’s lymphoma with a defined ratio of CD8+ and CD4+ CD19-specific chimeric antigen receptor-modified T cells, Sci Transl Med 8, 355ra116 (2016). - PMC - PubMed
-
- Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, Braunschweig I, Oluwole OO, Siddiqi T, Lin Y, Timmerman JM, Stiff PJ, Friedberg JW, Flinn IW, Goy A, Hill BT, Smith MR, Deol A, Farooq U, McSweeney P, Munoz J, Avivi I, Castro JE, Westin JR, Chavez JC, Ghobadi A, Komanduri KV, Levy R, Jacobsen ED, Witzig TE, Reagan P, Bot A, Rossi J, Navale L, Jiang Y, Aycock J, Elias M, Chang D, Wiezorek J, Go WY, Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma, N Engl J Med 377, 2531–2544 (2017). - PMC - PubMed
-
- Gardner RA, Finney O, Annesley C, Brakke H, Summers C, Leger K, Bleakley M, Brown C, Mgebroff S, Kelly-Spratt KS, Hoglund V, Lindgren C, Oron AP, Li D, Riddell SR, Park JR, Jensen MC, Intent-to-treat leukemia remission by CD19 CAR T cells of defined formulation and dose in children and young adults, Blood 129, 3322–3331 (2017). - PMC - PubMed
-
- Kochenderfer JN, Somerville RPT, Lu T, Shi V, Bot A, Rossi J, Xue A, Goff SL, Yang JC, Sherry RM, Klebanoff CA, Kammula US, Sherman M, Perez A, Yuan CM, Feldman T, Friedberg JW, Roschewski MJ, Feldman SA, McIntyre L, Toomey MA, Rosenberg SA, Lymphoma Remissions Caused by Anti-CD19 Chimeric Antigen Receptor T Cells Are Associated With High Serum Interleukin-15 Levels, J. Clin. Oncol, JCO2016713024 (2017). - PMC - PubMed
Publication types
- Comparative Study Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Research Support, N.I.H., Extramural Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Research Support, Non-U.S. Gov't Actions
- Search in PubMed
- Search in MeSH
- Add to Search
MeSH terms
- Humans Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Immunotherapy, Adoptive Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Multiple Myeloma* / therapy Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Receptors, Antigen, T-Cell / genetics Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Receptors, Chimeric Antigen* / genetics Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Signal Transduction Actions
- Search in PubMed
- Search in MeSH
- Add to Search
Substances
- Receptors, Antigen, T-Cell Actions
- Search in PubMed
- Search in MeSH
- Add to Search
- Receptors, Chimeric Antigen Actions
- Search in PubMed
- Search in MeSH
- Add to Search
Grants and funding
- R21 CA226962/CA/NCI NIH HHS/United States
- R50 CA211499/CA/NCI NIH HHS/United States
- R01 CA114536/CA/NCI NIH HHS/United States
- R01 CA136551/CA/NCI NIH HHS/United States
- U01 CA214114/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
- Atypon
- Europe PubMed Central
- PubMed Central
Other Literature Sources
- The Lens - Patent Citations Database
Medical
- MedlinePlus Health Information
Research Materials
- NCI CPTC Antibody Characterization Program
Miscellaneous
- NCI CPTAC Assay Portal
Tag » What Is Tcr In A Car
-
FEATURE: TCR Touring Car Platform Explained
-
TCR Touring Car - Wikipedia
-
Inside TCR - Hyundai Motorsport Official Website
-
What His TCR | We Help You Get The Fact About TCR Race Cars
-
A 'Brief' History Of TCR: Touring Car Specification - Petrolicious
-
TCR
-
CARS - TCR HUB
-
TCR HUB: HOME
-
What Is CAR T Cell, TCR Therapy, And What Are They Used For?
-
TCR-like CARs And TCR-CARs Targeting Neoepitopes - Nature
-
TCR Australia: Home
-
Guide To The TCR Australia 2022 Touring Car Racing Series
-
CUPRA TCR
-
Everything You Need To Know About TCR Australia - WhichCar