Dipole: Meaning, Examples & Types | StudySmarter
Maybe your like
Learning Materials
-
Explanations Explanations
- Anthropology
- Archaeology
- Architecture
- Art and Design
- Bengali
- Biology
- Business Studies
- Chemistry
- Chinese
- Combined Science
- Computer Science
- Economics
- Engineering
- English
- English Literature
- Environmental Science
- French
- Geography
- German
- Greek
- History
- Hospitality and Tourism
- Human Geography
- Japanese
- Italian
- Law
- Macroeconomics
- Marketing
- Math
- Media Studies
- Medicine
- Microeconomics
- Music
- Nursing
- Nutrition and Food Science
- Physics
- Politics
- Polish
- Psychology
- Religious Studies
- Sociology
- Spanish
- Sports Sciences
- Translation
Features
-
Flashcards
-
StudySmarter AI
-
Notes
-
Study Plans
-
Study Sets
-
Exams
Discover
-
Find a job
-
Student Deals
-
Magazine
-
Mobile App
- Explanations
- Chemistry
- Physical Chemistry
- Dipole Chemistry
Up until now, you have probably heard that water has many cool properties such as being polar, having cohesive and adhesive forces, and being a great solvent! But, what you ever heard about water being a dipole and wondered what exactly that means? If your answer is yes, you came to the right place!
Get startedMillions of flashcards designed to help you ace your studies
Sign up for free- + Add tag
- Immunology
- Cell Biology
- Mo
A _____ is a molecule that has a separation of charges.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
True or false: Dipoles occur when electrons are shared unequally between atoms due to a high difference in the electronegativity of the atoms involved.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
The formation of a dipole depends on the polarity of a bond, which is determined by the difference in ______ between the two atoms involved in the bond.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
_____ is the ability of an atom to attract electrons to itself.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
In ______ bonds, the electrons are equally shared between atoms.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
In ______, the electrons are shared unequally between atoms.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
______ do not have dipoles because they cancel out due to symmetry.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
In polar covalent bonds, dipoles are _____ present.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
True or false: Electronegativity decreases from left to right and decreases down a group.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
If the difference in electronegativity is _____ then the molecule will a non-polar covalent bond
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
If the difference in electronegativity is ______ then the molecule will have a polar covalent bond
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
A _____ is a molecule that has a separation of charges.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
True or false: Dipoles occur when electrons are shared unequally between atoms due to a high difference in the electronegativity of the atoms involved.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
The formation of a dipole depends on the polarity of a bond, which is determined by the difference in ______ between the two atoms involved in the bond.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
_____ is the ability of an atom to attract electrons to itself.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
In ______ bonds, the electrons are equally shared between atoms.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
In ______, the electrons are shared unequally between atoms.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
______ do not have dipoles because they cancel out due to symmetry.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
In polar covalent bonds, dipoles are _____ present.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
True or false: Electronegativity decreases from left to right and decreases down a group.
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
If the difference in electronegativity is _____ then the molecule will a non-polar covalent bond
Show Answer- + Add tag
- Immunology
- Cell Biology
- Mo
If the difference in electronegativity is ______ then the molecule will have a polar covalent bond
Show Answer Generate flashcards
Generate flashcards  Summarize page
Summarize page  Generate flashcards from highlight
Generate flashcards from highlight Review generated flashcards
Sign up for freeto start learning or create your own AI flashcards
Sign up for free You have reached the daily AI limitStart learning or create your own AI flashcards
 StudySmarter Editorial Team
StudySmarter Editorial Team Team Dipole Chemistry Teachers
- 10 minutes reading time
- Checked by StudySmarter Editorial Team
- Fact Checked Content
- Last Updated: 12.11.2022
- Published at: 26.05.2022
- 10 min reading time
- Chemical Analysis
- Chemical Reactions
- Chemistry Branches
- Inorganic Chemistry
- Ionic and Molecular Compounds
- Kinetics
- Making Measurements
- Nuclear Chemistry
- Organic Chemistry
- Physical Chemistry
- Absolute Entropy And Entropy Change
- Acid Dissociation Constant
- Acid-Base Indicators
- Acid-Base Reactions and Buffers
- Acids and Bases
- Alkali Metals
- Allotropes of Carbon
- Amorphous Polymer
- Amount of Substance
- Application of Le Chatelier's Principle
- Arrhenius Equation
- Arrhenius Theory
- Atom Economy
- Atomic Structure
- Autoionization of Water
- Avogadro Constant
- Avogadro's Number and the Mole
- Beer-Lambert Law
- Bond Enthalpy
- Bonding
- Born Haber Cycles
- Born-Haber Cycles Calculations
- Boyle's Law
- Brønsted-Lowry Acids and Bases
- Buffer Capacity
- Buffer Solutions
- Buffers
- Buffers Preparation
- Calculating Enthalpy Change
- Calculating Equilibrium Constant
- Calorimetry
- Carbon Structures
- Cell Potential
- Cell Potential and Free Energy
- Chalcogens
- Chemical Calculations
- Chemical Equations
- Chemical Equilibrium
- Chemical Thermodynamics
- Closed Systems
- Colligative Properties
- Collision Theory
- Common-Ion Effect
- Composite Materials
- Composition of Mixture
- Constant Pressure Calorimetry
- Constant Volume Calorimetry
- Coordination Compounds
- Coupling Reactions
- Covalent Bond
- Covalent Network Solid
- Crystalline Polymer
- De Broglie Wavelength
- Determining Rate Constant
- Deviation From Ideal Gas Law
- Diagonal Relationship
- Diamond
- Dilution
- Dipole Chemistry
- Dipole Moment
- Dissociation Constant
- Distillation
- Dynamic Equilibrium
- Electric Fields Chemistry
- Electrochemical Cell
- Electrochemical Series
- Electrochemistry
- Electrode Potential
- Electrolysis
- Electrolytes
- Electromagnetic Spectrum
- Electron Affinity
- Electron Configuration
- Electron Shells
- Electronegativity
- Electronic Transitions
- Elemental Analysis
- Elemental Composition of Pure Substances
- Empirical and Molecular Formula
- Endothermic and Exothermic Processes
- Energetics
- Energy Diagrams
- Enthalpy Changes
- Enthalpy For Phase Changes
- Enthalpy of Formation
- Enthalpy of Reaction
- Enthalpy of Solution and Hydration
- Entropy
- Entropy Change
- Equilibrium Concentrations
- Equilibrium Constant Kp
- Equilibrium Constants
- Examples of Covalent Bonding
- Factors Affecting Reaction Rates
- Finding Ka
- Free Energy
- Free Energy Of Dissolution
- Free Energy and Equilibrium
- Free Energy of Formation
- Fullerenes
- Fundamental Particles
- Galvanic and Electrolytic Cells
- Gas Constant
- Gas Solubility
- Gay Lussacs Law
- Giant Covalent Structures
- Graham's Law
- Graphite
- Ground State
- Group 3A
- Group 4A
- Group 5A
- Half Equations
- Heating Curve for Water
- Heisenberg Uncertainty Principle
- Henderson-Hasselbalch Equation
- Hess' Law
- Hybrid Orbitals
- Hydrogen Bonds
- Ideal Gas Law
- Ideal and Real Gases
- Intermolecular Forces
- Introduction to Acids and Bases
- Ion And Atom Photoelectron Spectroscopy
- Ion dipole Forces
- Ionic Bonding
- Ionic Product of Water
- Ionic Solids
- Ionisation Energy
- Ions: Anions and Cations
- Isotopes
- Kinetic Molecular Theory
- Lattice Structures
- Law of Definite Proportions
- Le Chatelier's Principle
- Lewis Acid and Bases
- London Dispersion Forces
- Magnitude Of Equilibrium Constant
- Mass Spectrometry
- Mass Spectrometry of Elements
- Maxwell-Boltzmann Distribution
- Measuring EMF
- Mechanisms of Chemical Bonding
- Melting and Boiling Point
- Metallic Bonding
- Metallic Solids
- Metals Non-Metals and Metalloids
- Mixtures and Solutions
- Molar Mass Calculations
- Molarity
- Molecular Orbital Theory
- Molecular Solid
- Molecular Structures of Acids and Bases
- Moles and Molar Mass
- Nanoparticles
- Neutralisation Reaction
- Oxidation Number
- Partial Pressure
- Particulate Model
- Partition Coefficient
- Percentage Yield
- Periodic Table Organization
- Phase Changes
- Phase Diagram of Water
- Photoelectric Effect
- Photoelectron Spectroscopy
- Physical Properties
- Polarity
- Polyatomic Ions
- Polyprotic Acid Titration
- Prediction of Element Properties Based on Periodic Trends
- Pressure and Density
- Properties Of Equilibrium Constant
- Properties of Buffers
- Properties of Solids
- Properties of Water
- Quantitative Electrolysis
- Quantum Energy
- Quantum Numbers
- RICE Tables
- Rate Equations
- Rate of Reaction and Temperature
- Reacting Masses
- Reaction Quotient
- Reaction Quotient And Le Chateliers Principle
- Real Gas
- Redox
- Relative Atomic Mass
- Representations of Equilibrium
- Reversible Reaction
- SI units chemistry
- Saturated Unsaturated and Supersaturated
- Shapes of Molecules
- Shielding Effect
- Simple Molecules
- Solids Liquids and Gases
- Solubility
- Solubility Curve
- Solubility Equilibria
- Solubility Product
- Solubility Product Calculations
- Solutes Solvents and Solutions
- Solution Representations
- Solutions and Mixtures
- Specific Heat
- Spectroscopy
- Standard Potential
- States of Matter
- Stoichiometry In Reactions
- Strength of Intermolecular Forces
- The Laws of Thermodynamics
- The Molar Volume of a Gas
- Thermodynamically Favored
- Trends in Ionic Charge
- Trends in Ionisation Energy
- Types of Mixtures
- VSEPR Theory
- Valence Electrons
- Van der Waals Forces
- Vapor Pressure
- Water in Chemical Reactions
- Wave Mechanical Model
- Weak Acid and Base Equilibria
- Weak Acids and Bases
- Writing Chemical Formulae
- pH
- pH Change
- pH Curves and Titrations
- pH Scale
- pH and Solubility
- pH and pKa
- pH and pOH
- The Earths Atmosphere
- Chemical Analysis
- Chemical Reactions
- Chemistry Branches
- Inorganic Chemistry
- Ionic and Molecular Compounds
- Kinetics
- Making Measurements
- Nuclear Chemistry
- Organic Chemistry
- Physical Chemistry
- Absolute Entropy And Entropy Change
- Acid Dissociation Constant
- Acid-Base Indicators
- Acid-Base Reactions and Buffers
- Acids and Bases
- Alkali Metals
- Allotropes of Carbon
- Amorphous Polymer
- Amount of Substance
- Application of Le Chatelier's Principle
- Arrhenius Equation
- Arrhenius Theory
- Atom Economy
- Atomic Structure
- Autoionization of Water
- Avogadro Constant
- Avogadro's Number and the Mole
- Beer-Lambert Law
- Bond Enthalpy
- Bonding
- Born Haber Cycles
- Born-Haber Cycles Calculations
- Boyle's Law
- Brønsted-Lowry Acids and Bases
- Buffer Capacity
- Buffer Solutions
- Buffers
- Buffers Preparation
- Calculating Enthalpy Change
- Calculating Equilibrium Constant
- Calorimetry
- Carbon Structures
- Cell Potential
- Cell Potential and Free Energy
- Chalcogens
- Chemical Calculations
- Chemical Equations
- Chemical Equilibrium
- Chemical Thermodynamics
- Closed Systems
- Colligative Properties
- Collision Theory
- Common-Ion Effect
- Composite Materials
- Composition of Mixture
- Constant Pressure Calorimetry
- Constant Volume Calorimetry
- Coordination Compounds
- Coupling Reactions
- Covalent Bond
- Covalent Network Solid
- Crystalline Polymer
- De Broglie Wavelength
- Determining Rate Constant
- Deviation From Ideal Gas Law
- Diagonal Relationship
- Diamond
- Dilution
- Dipole Chemistry
- Dipole Moment
- Dissociation Constant
- Distillation
- Dynamic Equilibrium
- Electric Fields Chemistry
- Electrochemical Cell
- Electrochemical Series
- Electrochemistry
- Electrode Potential
- Electrolysis
- Electrolytes
- Electromagnetic Spectrum
- Electron Affinity
- Electron Configuration
- Electron Shells
- Electronegativity
- Electronic Transitions
- Elemental Analysis
- Elemental Composition of Pure Substances
- Empirical and Molecular Formula
- Endothermic and Exothermic Processes
- Energetics
- Energy Diagrams
- Enthalpy Changes
- Enthalpy For Phase Changes
- Enthalpy of Formation
- Enthalpy of Reaction
- Enthalpy of Solution and Hydration
- Entropy
- Entropy Change
- Equilibrium Concentrations
- Equilibrium Constant Kp
- Equilibrium Constants
- Examples of Covalent Bonding
- Factors Affecting Reaction Rates
- Finding Ka
- Free Energy
- Free Energy Of Dissolution
- Free Energy and Equilibrium
- Free Energy of Formation
- Fullerenes
- Fundamental Particles
- Galvanic and Electrolytic Cells
- Gas Constant
- Gas Solubility
- Gay Lussacs Law
- Giant Covalent Structures
- Graham's Law
- Graphite
- Ground State
- Group 3A
- Group 4A
- Group 5A
- Half Equations
- Heating Curve for Water
- Heisenberg Uncertainty Principle
- Henderson-Hasselbalch Equation
- Hess' Law
- Hybrid Orbitals
- Hydrogen Bonds
- Ideal Gas Law
- Ideal and Real Gases
- Intermolecular Forces
- Introduction to Acids and Bases
- Ion And Atom Photoelectron Spectroscopy
- Ion dipole Forces
- Ionic Bonding
- Ionic Product of Water
- Ionic Solids
- Ionisation Energy
- Ions: Anions and Cations
- Isotopes
- Kinetic Molecular Theory
- Lattice Structures
- Law of Definite Proportions
- Le Chatelier's Principle
- Lewis Acid and Bases
- London Dispersion Forces
- Magnitude Of Equilibrium Constant
- Mass Spectrometry
- Mass Spectrometry of Elements
- Maxwell-Boltzmann Distribution
- Measuring EMF
- Mechanisms of Chemical Bonding
- Melting and Boiling Point
- Metallic Bonding
- Metallic Solids
- Metals Non-Metals and Metalloids
- Mixtures and Solutions
- Molar Mass Calculations
- Molarity
- Molecular Orbital Theory
- Molecular Solid
- Molecular Structures of Acids and Bases
- Moles and Molar Mass
- Nanoparticles
- Neutralisation Reaction
- Oxidation Number
- Partial Pressure
- Particulate Model
- Partition Coefficient
- Percentage Yield
- Periodic Table Organization
- Phase Changes
- Phase Diagram of Water
- Photoelectric Effect
- Photoelectron Spectroscopy
- Physical Properties
- Polarity
- Polyatomic Ions
- Polyprotic Acid Titration
- Prediction of Element Properties Based on Periodic Trends
- Pressure and Density
- Properties Of Equilibrium Constant
- Properties of Buffers
- Properties of Solids
- Properties of Water
- Quantitative Electrolysis
- Quantum Energy
- Quantum Numbers
- RICE Tables
- Rate Equations
- Rate of Reaction and Temperature
- Reacting Masses
- Reaction Quotient
- Reaction Quotient And Le Chateliers Principle
- Real Gas
- Redox
- Relative Atomic Mass
- Representations of Equilibrium
- Reversible Reaction
- SI units chemistry
- Saturated Unsaturated and Supersaturated
- Shapes of Molecules
- Shielding Effect
- Simple Molecules
- Solids Liquids and Gases
- Solubility
- Solubility Curve
- Solubility Equilibria
- Solubility Product
- Solubility Product Calculations
- Solutes Solvents and Solutions
- Solution Representations
- Solutions and Mixtures
- Specific Heat
- Spectroscopy
- Standard Potential
- States of Matter
- Stoichiometry In Reactions
- Strength of Intermolecular Forces
- The Laws of Thermodynamics
- The Molar Volume of a Gas
- Thermodynamically Favored
- Trends in Ionic Charge
- Trends in Ionisation Energy
- Types of Mixtures
- VSEPR Theory
- Valence Electrons
- Van der Waals Forces
- Vapor Pressure
- Water in Chemical Reactions
- Wave Mechanical Model
- Weak Acid and Base Equilibria
- Weak Acids and Bases
- Writing Chemical Formulae
- pH
- pH Change
- pH Curves and Titrations
- pH Scale
- pH and Solubility
- pH and pKa
- pH and pOH
- The Earths Atmosphere
- Fact Checked Content
- Last Updated: 12.11.2022
- 10 min reading time
- Content creation process designed by
 Lily Hulatt
Lily Hulatt - Content sources cross-checked by
 Gabriel Freitas
Gabriel Freitas - Content quality checked by
 Gabriel Freitas
Gabriel Freitas
Jump to a key chapter
- First, we will talk about the definition of a dipole and how dipoles are formed.
- Then, we will dive into the different types of dipoles in chemistry and give some examples.
Dipole Definition in Chemistry
Dipoles occur when electrons are shared unequally between atoms in the same molecule due to a high difference in the electronegativity of the atoms involved.
A dipole is a molecule or covalent bond that has a separation of charges.
Determination and Formation of a Dipole
The formation of a dipole depends on the polarity of a bond, which is determined by the difference in electronegativity between the two atoms involved in the bond.
Electronegativity is the ability of an atom to attract electrons to itself.
Types of Bonds
The three types of bonds you should be familiar with are non-polar covalent bonds, polar covalent bonds, and ionic bonds.
In non-polar covalent bonds, the electrons are equally shared between atoms. In polar covalent bonds, the electrons are shared unequally between atoms. In ionic bonds, the electrons are transferred.
- In ionic bonds, there are no dipoles.
- In polar covalent bonds, dipoles are always present.
- Non-polar covalent bonds do have dipoles but they cancel out due to symmetry.
Predicting Bond Polarity
To determine whether a bond is nonpolar covalent, polar covalent, or ionic, we need to look at the electronegativity values of the atoms involved and calculate the difference between them.
- If the difference in electronegativity is less than 0.4 → non-polar covalent bond
- If the difference in electronegativity falls between 0.4 and 1.7 → polar covalent bond
- If the difference in electronegativity is greater than 1.7 → ionic bond
The electronegativity values are given by Pauling's scale of electronegativity. In the periodic table below, we can see the electronegativity values for each element. Notice the trend here: electronegativity increases from left to right and decreases down a group.
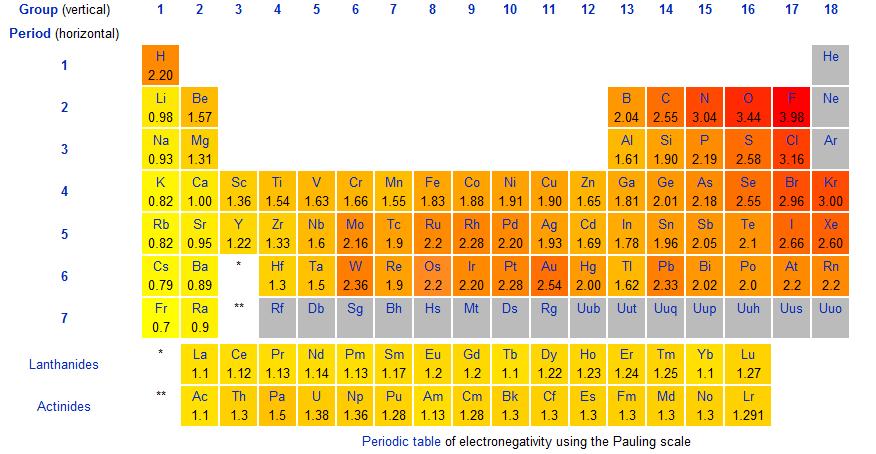
Fig.1-Periodic table showing Pauling's scale of electronegativity
Let's look at an example!
Predict the type of bond polarity between the following atoms:
a) H and Br
H has an EN value of 2.20 and Br has an EN of 2.96. The electronegativity difference between these atoms is 0.76 so it would have a polar covalent bond.
b) Li and F
Li has an EN value of 0.98 and F has an EN of 3.98. The electronegativity difference is 3.00 so it would have an ionic bond.
c) I and I
I has an EN value of 2.66. The electronegativity difference is 0.00 so it would have anon-polar covalent bond.
Dipole Moment in Chemistry
To measure the separation of charges in a molecule we use dipole moment. Dipole moments are present in polar molecules that have asymmetric shapes because, in asymmetric shapes, the dipoles do not cancel out.
Dipole moment is referred to as a measurement of the magnitude of a dipole.
To show the dipole moment, we use arrows pointing toward the more electronegative element. For example, in the figure below we can see an HCl and an SO3 molecule.
- In HCl, chlorine has a higher electronegativity value compared to hydrogen. So, the chlorine will have a partial negative charge and the hydrogen will have a partial positive charge. Since chlorine is more electronegative, the dipole arrow will point towards chlorine.
- In SO3, the oxygen atom has an electronegativity value higher than that of the sulfur atoms. So, the sulfur atom will have a partial positive charge and the oxygen atoms will have a partial negative charge. In this molecule, the symmetry causes the dipoles to cancel each other out. So, SO3 has no dipole moment.
Dipole moment of a bond can be calculated by using the following equation: μ=Q*r→ where Q is the magnitude of the partial charges δ+ and δ- , and r is the distance vector between the two charges. You can think of the distance vector as an arrow with pointing to the more electron-negative element from the less electron negative one. Dipole moment is measured in Debye units (D). The bigger the dipole moment of the bond, the more polar the bond is.
A dipole moment of a molecule is the sum of the dipole moments of the bonds. This is why it is important that we are using vectors. Vectors have a property called directionality, meaning they point from somewhere to somewhere. You see if two vectors are equally long and point in the opposite direction ( + and -) their sum will be zero. So in theory, if the molecule is perfectly symmetric, meaning all vectors will add up to 0 the dipole moment of the whole molecule will be zero. Okay, let's take a look at an example.
You can learn more about the different molecular shapes by reading "Valence Shell Electron Pair Repulsion (VSEPR) Theory.
Which of the following compounds has a dipole moment? PCl3 or PCl5?
First, we need to take a look at their lewis structures. If the structure is symmetrical, then the dipoles will cancel out and the compound will not have a dipole.
In PCl3, the bond is polar because of the difference in electronegativity between P and Cl atoms, and the presence of a lone pair of electrons gives PCl3 a tetrahedral structure.
On the other hand, PCl5 is considered non-polar because its symmetrical shape, which is trigonal bipyramidal, cancels the dipoles out.
 Fig.2-Lewis diagrams of phosphorus trichloride and phosphorus pentachloride
Fig.2-Lewis diagrams of phosphorus trichloride and phosphorus pentachloride
If you need to go back and learn how to draw Lewis structures, check out "Lewis Diagrams".
Types of Dipole in Chemistry
The three types of dipole interactions you might encounter are called ion-dipole, dipole-dipole, and induced-dipole induced-dipole (London dispersion forces).
Ion-Dipole
An ion-dipole interaction occurs between an ion and a polar (dipole) molecule. The higher the ion charge, the stronger the ion-dipole attractive force is. An example of ion-dipole is sodium ion in water.
 Fig.3-Ion-dipole forces holding sodium ion and water
Fig.3-Ion-dipole forces holding sodium ion and water
Another type of interaction involving ions is ion-induced dipole force. This interaction occurs when a charged ion induces a temporary dipole in a non-polar molecule. For example, Fe3+ can induce a temporary dipole in O2, giving rise to an ion-induced dipole interaction!
So what does it mean to induce a dipole? If you put an ion near a non-polar molecule, you can start affecting its electrons. For example, a positive ion will attract these electrons to the side on which the ion is. This will create a larger concentration of ions there and lead to a dipole forming on the originally non-polar molecule.
Dipole-Dipole
When two polar molecules possessing permanent dipoles are near each other, attractive forces called dipole-dipole interactions hold the molecules together. Dipole-dipole interactions are attractive forces that occur between the positive end of a polar molecular and the negative end of another polar molecule. A common example of dipole-dipole forces is seen between HCl molecules. In HCl, the partial positive H atoms get attracted to the partial negative Cl atoms of another molecule.
 Fig.4-Dipole-dipole forces between HCl molecules
Fig.4-Dipole-dipole forces between HCl molecules
Hydrogen Bonding
A special type of dipole-dipole interaction is hydrogen bonding. Hydrogen bonding is an intermolecular force that occurs between the hydrogen atom covalently bonded to an N, O, or F and another molecule containing N, O, or F. For example, in water (H2O), the H atom covalently bonded to oxygen gets attracted to the oxygen of another water molecule, creating hydrogen bonding.
 Fig.5-Hydrogen bonding between water molecules
Fig.5-Hydrogen bonding between water molecules
Dipole-induced Dipole forces
Dipole-induced dipole forces arise when a polar molecule with a permanent dipole induces a temporary dipole in a non-polar molecule. For example, dipole-induced dipole forces can hold molecules of HCl and He atoms together.
London dispersion forces
Induced-dipole Induced-dipole interactions are also known as London dispersion forces. This type of interaction is present in all molecules, but it is most important when dealing with non-polar molecules. London dispersion forces occur because of the random movement of electrons in the cloud of electrons. This movement produces a weak, temporary dipole moment! For example, London dispersion forces are the only type of attractive force holding F2 molecules together.
Examples of Dipoles in Chemistry
Now that you have a better understanding of what dipoles are, let's look at more examples! If the figure below you can see the structure of acetone. Acetone, C3H6O, is a polar molecular with a bond dipole.
 Fig.6-Dipoles in Acetone
Fig.6-Dipoles in Acetone
Another common example of a molecule containing dipoles is carbon tetrachloride, CCl4. Carbon tetrachloride is a non-polar molecule that contains polar bonds, and therefore, has dipoles present. However, the net dipole is zero due to its tetrahedral structure, where the bond dipoles directly oppose each other.
 Fig.7-Structure of Carbon Tetrachloride
Fig.7-Structure of Carbon Tetrachloride
Let's look at one last example!
What is the net dipole moment in CO2?
CO2 is a linear molecule that has two C=O bond dipoles equal in magnitude but pointing in opposite directions. Therefore, the net dipole moment is zero.
 Fig.8-Dipoles in Carbon Dioxide
Fig.8-Dipoles in Carbon Dioxide
Dipoles can be a little intimidating, but once you get the hang of it you will find it simple!
Dipoles - Key takeaways
- Dipoles occur when electrons are shared unequally between atoms due to a high difference in the electronegativity of the atoms involved.
- A dipole moment is referred to as a measurement of the magnitude of a dipole.
- Dipole moments are present in polar molecules that have asymmetric shapes because, in asymmetric shapes, the dipoles do not cancel out.
- Types of dipoles include ion-dipole, dipole-dipole, and induced-dipole induced-dipole (London dispersion forces).
References:
Saunders, N. (2020). Supersimple Chemistry: The Ultimate Bitesize Study Guide. London: Dorling Kindersley.
Timberlake, K. C. (2019). Chemistry: An introduction to general, organic, and Biological Chemistry. New York, NY: Pearson.
Malone, L. J., Dolter, T. O., & Gentemann, S. (2013). Basic concepts of Chemistry (8th ed.). Hoboken, NJ: John Wiley & Sons.
Brown, T. L., LeMay, H. E., Bursten, B. E., Murphy, C. J., Woodward, P. M., Stoltzfus, M., & Lufaso, M. W. (2018). Chemistry: The central science (13th ed.). Harlow, United Kingdom: Pearson.
References
- Fig.1-Periodic table showing Pauling's scale of electronegativity (https://upload.wikimedia.org/wikipedia/commons/thumb/4/42/Electronegative.jpg/640px-Electronegative.jpg) by ad blocker on wikimedia commons licensed by CC By-SA 3.0 (https://creativecommons.org/licenses/by-sa/3.0/)
Similar topics in Chemistry
- Kinetics
- Chemistry Branches
- Ionic and Molecular Compounds
- Chemical Reactions
- Chemical Analysis
- Physical Chemistry
- Organic Chemistry
- Inorganic Chemistry
- Making Measurements
- Nuclear Chemistry
- The Earths Atmosphere
Related topics to Physical Chemistry
- Born-Haber Cycles Calculations
- Buffers Preparation
- Real Gas
- Metallic Bonding
- Born Haber Cycles
- Redox
- Vapor Pressure
- Carbon Structures
- Free Energy of Formation
- Ionic Product of Water
- pH Curves and Titrations
- Trends in Ionisation Energy
- Ground State
- Bond Enthalpy
- Chemical Thermodynamics
- Diagonal Relationship
- Cell Potential
- pH Change
- Solubility
- Equilibrium Constant Kp
- Molecular Structures of Acids and Bases
- Buffer Capacity
- Solids Liquids and Gases
- Chemical Equilibrium
- Free Energy
- Gas Constant
- Alkali Metals
- Ionisation Energy
- Relative Atomic Mass
- Measuring EMF
- Rate Equations
- Diamond
- Simple Molecules
- Collision Theory
- Energetics
- pH and pKa
- Reversible Reaction
- Calorimetry
- Molar Mass Calculations
- Avogadro Constant
- Fullerenes
- Partial Pressure
- Solution Representations
- Maxwell-Boltzmann Distribution
- Acid Dissociation Constant
- Autoionization of Water
- Molecular Solid
- Standard Potential
- Ionic Bonding
- Allotropes of Carbon
- Composite Materials
- Electronegativity
- Spectroscopy
- Ions: Anions and Cations
- Electrochemistry
- Common-Ion Effect
- Endothermic and Exothermic Processes
- Electron Shells
- Mechanisms of Chemical Bonding
- pH and pOH
- Law of Definite Proportions
- Cell Potential and Free Energy
- Van der Waals Forces
- Mixtures and Solutions
- Equilibrium Constants
- Dynamic Equilibrium
- Calculating Equilibrium Constant
- Chemical Calculations
- Hess' Law
- Factors Affecting Reaction Rates
- Molecular Orbital Theory
- Phase Changes
- Ideal and Real Gases
- Photoelectron Spectroscopy
- Electrode Potential
- Reacting Masses
- Constant Pressure Calorimetry
- Electrochemical Series
- Polyatomic Ions
- Gas Solubility
- Determining Rate Constant
- Buffer Solutions
- Solubility Product
- Solubility Equilibria
- Partition Coefficient
- Ideal Gas Law
- SI units chemistry
- Le Chatelier's Principle
- Electron Affinity
- Calculating Enthalpy Change
- Properties of Buffers
- Enthalpy of Reaction
- Properties of Water
- Molarity
- Moles and Molar Mass
- Entropy
- Strength of Intermolecular Forces
- Arrhenius Equation
- Examples of Covalent Bonding
- Pressure and Density
- Beer-Lambert Law
- VSEPR Theory
- Acid-Base Indicators
- Water in Chemical Reactions
- Electromagnetic Spectrum
- Properties of Solids
- Covalent Network Solid
- Particulate Model
- Metallic Solids
- Buffers
- Deviation From Ideal Gas Law
- Hydrogen Bonds
- Avogadro's Number and the Mole
- Application of Le Chatelier's Principle
- Kinetic Molecular Theory
- Distillation
- Electrochemical Cell
- Weak Acid and Base Equilibria
- Chalcogens
- Melting and Boiling Point
- Half Equations
- Mass Spectrometry of Elements
- Rate of Reaction and Temperature
- Enthalpy Changes
- Energy Diagrams
- Thermodynamically Favored
- pH and Solubility
- Solubility Product Calculations
- Phase Diagram of Water
- Solubility Curve
- Heisenberg Uncertainty Principle
- Amorphous Polymer
- Crystalline Polymer
- London Dispersion Forces
- Dissociation Constant
- Neutralisation Reaction
- Arrhenius Theory
- Oxidation Number
- Electric Fields Chemistry
- Dipole Moment
- Graphite
- Electronic Transitions
- Wave Mechanical Model
- Coupling Reactions
- Trends in Ionic Charge
- Quantum Energy
- Acid-Base Reactions and Buffers
- Quantitative Electrolysis
- De Broglie Wavelength
- Ionic Solids
- Boyle's Law
- Lattice Structures
- Dipole Chemistry
- pH Scale
- States of Matter
- Atomic Structure
- Fundamental Particles
- Isotopes
- Bonding
- Electron Configuration
- Covalent Bond
- Amount of Substance
- Empirical and Molecular Formula
- Percentage Yield
- Physical Properties
- Shapes of Molecules
- Polarity
- Intermolecular Forces
- Atom Economy
- Acids and Bases
- Brønsted-Lowry Acids and Bases
- pH
- Weak Acids and Bases
- Solutions and Mixtures
- Reaction Quotient
- Giant Covalent Structures
- Elemental Composition of Pure Substances
- Equilibrium Concentrations
- The Laws of Thermodynamics
- The Molar Volume of a Gas
- Ion dipole Forces
- Shielding Effect
- Dilution
- Types of Mixtures
- Writing Chemical Formulae
- Heating Curve for Water
- Photoelectric Effect
- Henderson-Hasselbalch Equation
- Valence Electrons
- Composition of Mixture
- Galvanic and Electrolytic Cells
- Elemental Analysis
- Finding Ka
- Enthalpy of Solution and Hydration
- Entropy Change
- Graham's Law
- RICE Tables
- Prediction of Element Properties Based on Periodic Trends
- Solutes Solvents and Solutions
- Quantum Numbers
- Specific Heat
- Colligative Properties
- Enthalpy of Formation
- Group 3A
- Introduction to Acids and Bases
- Electrolytes
- Absolute Entropy And Entropy Change
- Group 4A
- Saturated Unsaturated and Supersaturated
- Group 5A
- Closed Systems
- Lewis Acid and Bases
- Metals Non-Metals and Metalloids
- Chemical Equations
- Coordination Compounds
- Nanoparticles
- Stoichiometry In Reactions
- Electrolysis
- Enthalpy For Phase Changes
- Free Energy and Equilibrium
- Mass Spectrometry
- Magnitude Of Equilibrium Constant
- Polyprotic Acid Titration
- Hybrid Orbitals
- Properties Of Equilibrium Constant
- Reaction Quotient And Le Chateliers Principle
- Constant Volume Calorimetry
- Free Energy Of Dissolution
- Gay Lussacs Law
- Periodic Table Organization
- Ion And Atom Photoelectron Spectroscopy
- Representations of Equilibrium
Flashcards in Dipole Chemistry
19 Start learningA _____ is a molecule that has a separation of charges.
Dipole
True or false: Dipoles occur when electrons are shared unequally between atoms due to a high difference in the electronegativity of the atoms involved.
True
The formation of a dipole depends on the polarity of a bond, which is determined by the difference in ______ between the two atoms involved in the bond.
Electronegativity
_____ is the ability of an atom to attract electrons to itself.
Electronegativity
In ______ bonds, the electrons are equally shared between atoms.
non-polar covalent
In ______, the electrons are shared unequally between atoms.
polar covalent bonds
Learn faster with the 19 flashcards about Dipole Chemistry
Sign up for free to gain access to all our flashcards.
 Sign up with Email
Sign up with Email Already have an account? Log in
Frequently Asked Questions about Dipole Chemistry
How to calculate dipole moment?
Dipole moment can be calculated by using the following equation: = Qr where Q is the magnitude of the partial charges δ+ and δ- , and r is the distance between the two charges.
How do you determine a dipole?
The formation of a dipole depends on the polarity of a bond, which is determined by the difference in electronegativity between the two atoms involved in the bond.
What causes a dipole in chemistry?
Dipoles are caused when electrons are shared unequally between atoms due to a high difference in the electronegativity of the atoms involved.
What is a dipole moment in chemistry?
Dipole moment is referred to as a measurement of the magnitude of a dipole.
What is a dipole in chemistry?
A dipole is a molecule that has a separation of charges.
Save Article Test your knowledge with multiple choice flashcardsA _____ is a molecule that has a separation of charges.
A. Ionic bond B. Dipole moment C. DipoleTrue or false: Dipoles occur when electrons are shared unequally between atoms due to a high difference in the electronegativity of the atoms involved.
A. True B. FalseThe formation of a dipole depends on the polarity of a bond, which is determined by the difference in ______ between the two atoms involved in the bond.
A. Boiling point B. Polarity C. ElectronegativityScore
That was a fantastic start! You can do better! Sign up to create your own flashcardsAccess over 700 million learning materials
Study more efficiently with flashcards
Get better grades with AI
Sign up for freeAlready have an account? Log in
Good job!Keep learning, you are doing great.
Don't give up! Next Open in our appHow we ensure our content is accurate and trustworthy?
At StudySmarter, we have created a learning platform that serves millions of students. Meet the people who work hard to deliver fact based content as well as making sure it is verified.
Content Creation Process: Lily Hulatt
Lily Hulatt Digital Content Specialist
Lily Hulatt is a Digital Content Specialist with over three years of experience in content strategy and curriculum design. She gained her PhD in English Literature from Durham University in 2022, taught in Durham University’s English Studies Department, and has contributed to a number of publications. Lily specialises in English Literature, English Language, History, and Philosophy.
Get to know Lily Content Quality Monitored by: Gabriel Freitas
Gabriel Freitas AI Engineer
Gabriel Freitas is an AI Engineer with a solid experience in software development, machine learning algorithms, and generative AI, including large language models’ (LLMs) applications. Graduated in Electrical Engineering at the University of São Paulo, he is currently pursuing an MSc in Computer Engineering at the University of Campinas, specializing in machine learning topics. Gabriel has a strong background in software engineering and has worked on projects involving computer vision, embedded AI, and LLM applications.
Get to know Gabriel Discover learning materials with the free StudySmarter app Sign up for free

About StudySmarter
StudySmarter is a globally recognized educational technology company, offering a holistic learning platform designed for students of all ages and educational levels. Our platform provides learning support for a wide range of subjects, including STEM, Social Sciences, and Languages and also helps students to successfully master various tests and exams worldwide, such as GCSE, A Level, SAT, ACT, Abitur, and more. We offer an extensive library of learning materials, including interactive flashcards, comprehensive textbook solutions, and detailed explanations. The cutting-edge technology and tools we provide help students create their own learning materials. StudySmarter’s content is not only expert-verified but also regularly updated to ensure accuracy and relevance.
Learn more StudySmarter Editorial Team
StudySmarter Editorial Team Team Chemistry Teachers
- 10 minutes reading time
- Checked by StudySmarter Editorial Team
Save explanations to your personalised space and access them anytime, anywhere!
Sign up with Email Sign up with AppleBy signing up, you agree to the Terms and Conditions and the Privacy Policy of StudySmarter.
Already have an account? Log in
Sign up to highlight and take notes. It’s 100% free.
Get Started Free Explore our app and discover over 50 million learning materials for free. Sign up for free 94% of StudySmarter users achieve better grades with our free platform. Download now!Tag » What Is A Dipole Dipole Force
-
Dipole-Dipole Forces
-
Dipole–dipole Forces (video) | Khan Academy
-
Dipole-Dipole Interactions - Chemistry LibreTexts
-
Dipole-Dipole - Chemistry 301
-
Hydrogen Bonding, Dipole-Dipole & Ion-Dipole Forces
-
What Are Dipole Dipole Forces? - Chemistry Question - Byju's
-
Dipole Interaction - An Overview | ScienceDirect Topics
-
What Is A Dipole - Dipole Force? - Toppr
-
Intermolecular Force - Wikipedia
-
[PDF] TYPES OF INTERMOLECULAR FORCES
-
Dipole Dipole Forces Of Attraction - Intermolecular Forces - YouTube
-
Dipole Dipole Forces And Interactions | Chemistry - YouTube
-
A Level Chemistry Revision "Permanent Dipole-Dipole Interactions"
-
Dipole-dipole Forces: Definition And Examples - Chemistry Learner