Flame Tests - Chemistry LibreTexts
Maybe your like
The Origin of Flame Colors
If you excite an atom or an ion by very strong heating, electrons can be promoted from their normal unexcited state into higher orbitals. As they fall back down to lower levels (either in one go or in several steps), energy is released as light. Each of these jumps involves a specific amount of energy being released as light energy, and each corresponds to a particular wavelength (or frequency). As a result of all these jumps, a spectrum of lines will be produced, some of which will be in the visible part of the spectrum. The color you see will be a combination of all these individual colors.
In the case of sodium (or other metal) ions, the jumps involve very high energies and these result in lines in the UV part of the spectrum which your eyes can't see. The jumps that you can see in flame tests come from electrons falling from a higher to a lower level in the metal atoms. So if, for example, you put sodium chloride which contains sodium ions, into a flame, where do the atoms come from? In the hot flame, some of the sodium ions regain their electrons to form neutral sodium atoms again. A sodium atom in an unexcited state has the structure 1s22s22p63s1, but within the flame there will be all sorts of excited states of the electrons. Sodium's familiar bright orange-yellow flame color results from promoted electrons falling back from the 3p1 level to their normal 3s1 level.

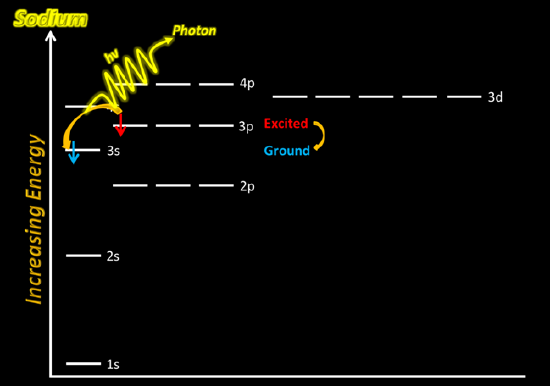
The exact sizes of the possible jumps in energy terms vary from one metal to another. That means that each different metal will have a different pattern of spectral lines, and so a different flame color. Flame colors are produced from the movement of the electrons in the metal ions present in the compounds. For example, a sodium ion in an unexcited state has the electron configuration 1s22s22p6. When heated, the electrons gain energy and can be excited into any of the empty higher-energy orbitals—7s, 6p, 4d, or any other, depending on the amount of energy a particular electron happens to absorb from the flame. Because the electron is now at a higher and more energetically unstable level, it falls back down to the original level, but not necessarily in one transition.
The electron transitions which produced lines in the visible spectrum involved atoms rather than ions.
Tag » What Color Does Sodium Burn
-
Sizzling Science: Exploring The Chemistry Of Fireworks
-
If You Burn Sodium, What Colour Flame Will Be Produced? - Quora
-
Flame Tests | Causes Of Color - Webexhibits
-
Flame Test Colors: Photo Gallery - ThoughtCo
-
Flame Test - Wikipedia
-
Sodium Salt Shows Which Of The Colour In A Flame Test? - Toppr
-
Sodium Flame Test - YouTube
-
Sodium Flame Color - YouTube
-
TOP 9 What Color Does Sodium Burn BEST And NEWEST
-
Colors Of Elements In A Flame - Sodium Chloride - ChemEdX
-
Lesson Explainer: Flame Tests - Nagwa
-
Flame Tests Using Metal Salts | Resource - RSC Education
-
How Fireworks Get Their Colors - Science Friday
-
Classroom Resources | Flame Test (Rainbow Demo) - AACT