Oxidizing And Reducing Agents - Chemistry LibreTexts
Maybe your like
Oxidizing and Reducing Agents
An oxidizing agent, or oxidant, gains electrons and is reduced in a chemical reaction. Also known as the electron acceptor, the oxidizing agent is normally in one of its higher possible oxidation states because it will gain electrons and be reduced. Examples of oxidizing agents include halogens, potassium nitrate, and nitric acid.
A reducing agent, or reductant, loses electrons and is oxidized in a chemical reaction. A reducing agent is typically in one of its lower possible oxidation states, and is known as the electron donor. A reducing agent is oxidized, because it loses electrons in the redox reaction. Examples of reducing agents include the earth metals, formic acid, and sulfite compounds.
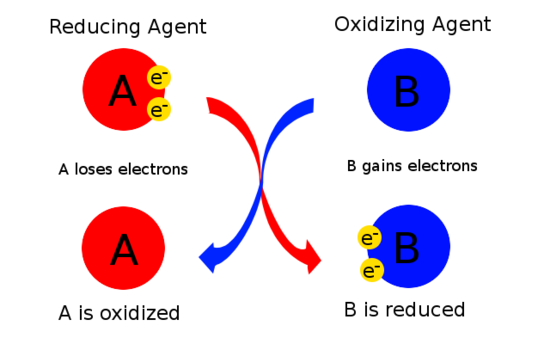
To help eliminate confusion, there is a mnemonic device to help determine oxidizing and reducing agents.
OIL RIG:
Oxidation Is Loss and Reduction Is Gain of electrons
| Common oxidizing agents | Common reducing agents |
|---|---|
| \(\ce{O2}\) | \(\ce{H2}\) |
| \(\ce{O3}\) | \(\ce{CO}\) |
| \(\ce{F2}\) | \(\ce{Fe}\) |
| \(\ce{Br2}\) | \(\ce{Zn}\) |
| \(\ce{H2SO4}\) | \(\ce{Li}\) |
| Halogens (they favor gaining an electron to obtain a noble gas configuration) | Alkali metals (they favor losing an electron to obtain a noble gas configuration) |
Example \(\PageIndex{1}\): Identify Reducing and Oxidizing Agents
Identify the reducing and oxidizing agents in the balanced redox reaction:
\[\ce{Cl2 (aq) + 2Br^{-} (aq) -> 2Cl^{-} (aq) + Br2 (aq)} \nonumber\]Oxidation half reaction
\[\ce{2 Br^{-} (aq) -> Br2 (aq)} \nonumber\] Oxidation States: -1 0Reduction Half Reaction
\[\ce{Cl2 (aq) -> 2 Cl^{-} (aq)} \nonumber\] Oxidation States: 0 -1Overview
- Br- loses an electron; it is oxidized from Br- to Br2; thus, Br- is the reducing agent.
- Cl2 gains one electron; it is reduced from Cl2 to 2 Cl-; thus, Cl2 is the oxidizing agent.
Exercise \(\PageIndex{1}\)
Identify the oxidizing agent and the reducing agent in the following redox reaction:
\[\ce{MnO4^{-} + SO3^{2-} -> Mn^{+2} + SO4^{2-}}\nonumber\]
Answer\(\ce{SO3^{2-}}\) is the reducing agent and \(\ce{MnO4^{-}}\) is the oxidizing agent. Note that while a specific atom typically has an odization state changes, the agents are the actual species, not the atoms.
Tag » What Is A Reducing Agent
-
Reducing Agent - Wikipedia
-
CHEBI:63247 - Reducing Agent - EMBL-EBI
-
What Is A Reducing Agent? - Definition From Corrosionpedia
-
Reducing Agents | Definition, Benefits & More | Overview
-
Reducing Agents - GoldBio
-
Reducing Agent
-
Illustrated Glossary Of Organic Chemistry - Reducing Agent
-
Oxidising And Reducing Agents - Higher Chemistry Revision - BBC
-
Reducing Agent Definition & Meaning - Merriam-Webster
-
Define Oxidising And Reducing Agents By Giving Suitable Example.
-
Reducing Agent | Chemistry - Britannica
-
Oxidizing Agents And Reducing Agents - YouTube