Relationships Among Pressure, Temperature, Volume, And Amount
Maybe your like
The Relationship between Pressure and Volume: Boyle's Law
As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Conversely, as the pressure on a gas decreases, the gas volume increases because the gas particles can now move farther apart. Weather balloons get larger as they rise through the atmosphere to regions of lower pressure because the volume of the gas has increased; that is, the atmospheric gas exerts less pressure on the surface of the balloon, so the interior gas expands until the internal and external pressures are equal.
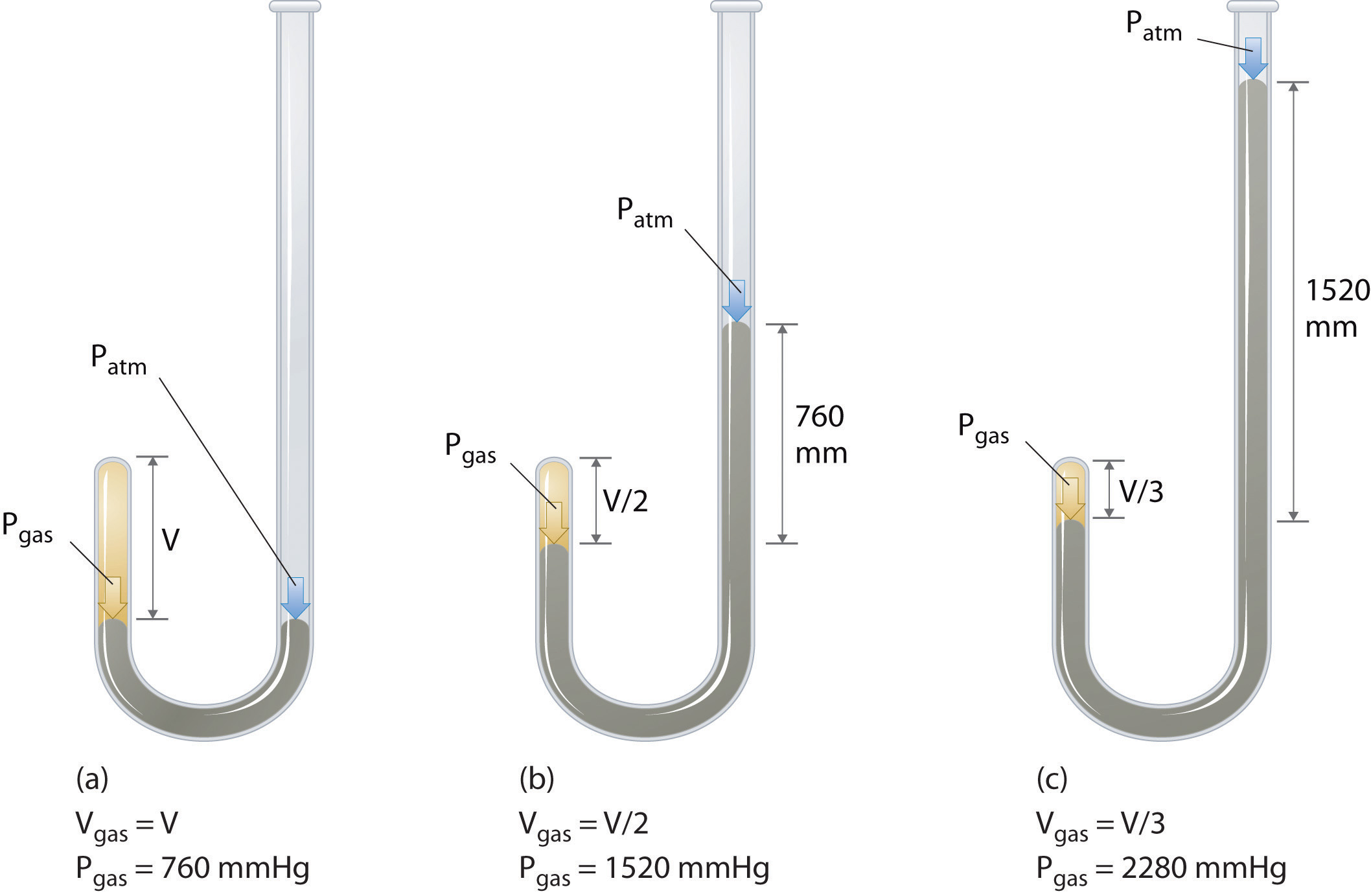
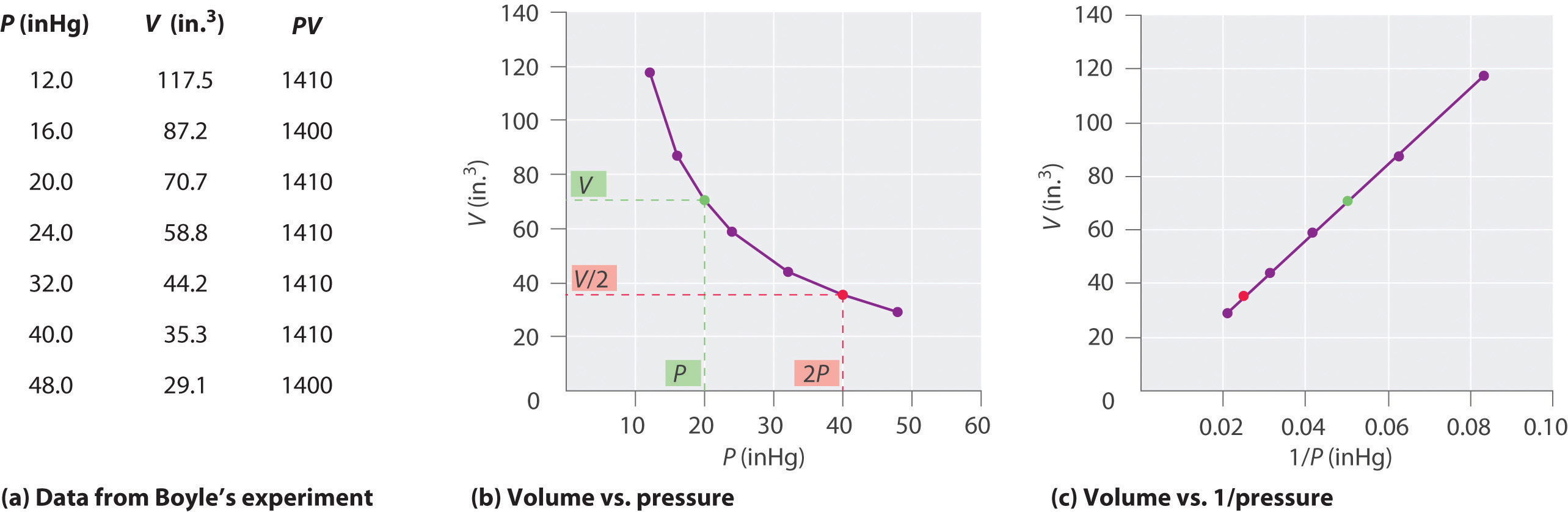
The Irish chemist Robert Boyle (1627–1691) carried out some of the earliest experiments that determined the quantitative relationship between the pressure and the volume of a gas. Boyle used a J-shaped tube partially filled with mercury, as shown in Figure \(\PageIndex{1}\). In these experiments, a small amount of a gas or air is trapped above the mercury column, and its volume is measured at atmospheric pressure and constant temperature. More mercury is then poured into the open arm to increase the pressure on the gas sample. The pressure on the gas is atmospheric pressure plus the difference in the heights of the mercury columns, and the resulting volume is measured. This process is repeated until either there is no more room in the open arm or the volume of the gas is too small to be measured accurately. Data such as those from one of Boyle’s own experiments may be plotted in several ways (Figure \(\PageIndex{2}\)). A simple plot of \(V\) versus \(P\) gives a curve called a hyperbola and reveals an inverse relationship between pressure and volume: as the pressure is doubled, the volume decreases by a factor of two. This relationship between the two quantities is described as follows:
\[PV = \rm constant \label{6.2.1}\]
Dividing both sides by \(P\) gives an equation illustrating the inverse relationship between \(P\) and \(V\):
\[V=\dfrac{\rm const.}{P} = {\rm const.}\left(\dfrac{1}{P}\right) \label{6.2.2}\]
or
\[V \propto \dfrac{1}{P} \label{6.2.3}\]
where the ∝ symbol is read “is proportional to.” A plot of V versus 1/P is thus a straight line whose slope is equal to the constant in Equation 6.2.1 and Equation 6.2.3. Dividing both sides of Equation 6.2.1 by V instead of P gives a similar relationship between P and 1/V. The numerical value of the constant depends on the amount of gas used in the experiment and on the temperature at which the experiments are carried out. This relationship between pressure and volume is known as Boyle’s law, after its discoverer, and can be stated as follows: At constant temperature, the volume of a fixed amount of a gas is inversely proportional to its pressure.
Boyle’s Law: https://youtu.be/lu86VSupPO4
Tag » When Volume Decreases What Happens To Pressure
-
Volume And Pressure In Gases – The Gas Laws - BBC Bitesize
-
If Volume Decreases In A Gas, What Happens To Pressure? - Socratic
-
What Happens To Pressure If Volume Decreases In A Gas? - Quora
-
Why Does Pressure Increase When Volume Decreases - Byju's
-
Gas Laws
-
Why Does The Pressure Decrease As Volume Increases? - Sciencing
-
Boyle's Law - Wikipedia
-
9.2 Relating Pressure, Volume, Amount, And Temperature: The Ideal ...
-
What Happens When The Pressure & Temperature Of A Fixed ...
-
What Happens To The Pressure When The Volume Is Decreased? - Toppr
-
Ideal Gas Law
-
When Volume Decreases What Happens To Pressure? - FAQ Blog
-
Boyle's Law | Science Primer
-
Boyle's Law | Understanding Gas Laws - ChemTalk