2.10: Organic Acids And Organic Bases - Chemistry LibreTexts
Có thể bạn quan tâm
Inductive Effects in Nitrogen Basicity
Alkylamines are more basic than ammonia since alkyl groups donate electrons to the more electronegative nitrogen. This inductive effect makes the electron density on the alkylamine nitrogen greater than the nitrogen of ammonium. That means that there will be a small amount of extra negative charge built up on the nitrogen atom. That extra negativity around the nitrogen makes the lone pair even more attractive towards hydrogen ions. Correspondingly, primary, secondary, and tertiary alkyl amines are more basic than ammonia.
methyl group pushes electron density toward the nitrogen, making it more basic
Making the nitrogen more negative helps the lone pair to pick up a hydrogen ion. What about the effect on the positive methylammonium ion formed? Is this more stable than a simple ammonium ion? Compare the methylammonium ion with an ammonium ion:
In the methylammonium ion, the positive charge is spread around the ion by the "electron-pushing" effect of the methyl group. The more you can spread charge around, the more stable an ion becomes. In the ammonium ion there is not any way of spreading the charge.
To summarize:
- The nitrogen is more negative in methylamine than in ammonia, and so it picks up a hydrogen ion more readily.
- The ion formed from methylamine is more stable than the one formed from ammonia, and so is less likely to shed the hydrogen ion again.
Taken together, these mean that methylamine is a stronger base than ammonia.
| Compound | pKa |
| NH3 | 9.3 |
| CH3NH2 | 10.66 |
| (CH3)2NH | 10.74 |
| (CH3)3N | 9.81 |
Resonance Effects in Nitrogen Basicity
The resonance effect also explains why a nitrogen atom is basic when it is in an amine, but not significantly basic when it is part of an amide group. While the lone pair of electrons in an amine nitrogen is localized in one place, the lone pair on an amide nitrogen is delocalized by resonance. The lone pair is stabilized by resonance delocalization. Here’s another way to think about it: the lone pair on an amide nitrogen is not available for bonding with a proton – these two electrons are too stable being part of the delocalized pi-bonding system. The electrostatic potential map show the effect of resonance on the basicity of an amide. The map shows that the electron density, shown in red, is almost completely shifted towards the oxygen. This greatly decreases the basicity of the lone pair electrons on the nitrogen in an amide.
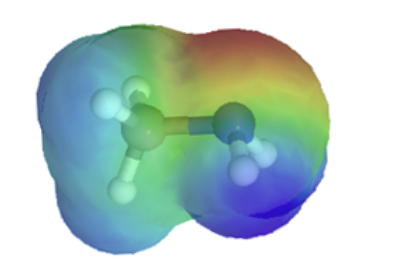 | 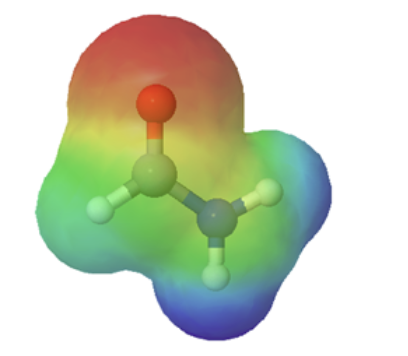 |
Aniline, the amine analog of phenol, is substantially less basic than an amine (as evidenced by the pKa of the conjugate acids).
|
|
We can use the same reasoning that we used when comparing the acidity of a phenol to that of an alcohol. In aniline, the lone pair on the nitrogen atom is stabilized by resonance with the aromatic pi system, making it less available for bonding and thus less basic.
lone pair is stabilized through resonance
In these cases, you seem to be breaking the same oxygen-hydrogen bond each time, and so you might expect the strengths to be similar. The most important factor in determining the relative acid strengths of these molecules is the nature of the ions formed. You always get a hydronium ion - so that's constant - but the nature of the anion (the negative ion) varies markedly from case to case.
Từ khóa » Ch3nh2 Pka
-
Methylamine, Also Known As Aminomethane Or Methanamine, Belongs To The Class Of Organic Compounds Known As Monoalkylamines. ... Structure For FDB003958 (Methylamine)
-
Methylamine | CH3NH2 - PubChem
-
How Do You Find The PKa Of CH3NH2? - Quora
-
What Is The PKa Of Methylamine - Chemistry Stack Exchange
-
Methylamine - Wikipedia
-
Pka Of Amines - Chemical Forums
-
Amine Basicity Is Measued By The PKa Of Its Conjugate Acid (pKaH)
-
Methylamine: Uses, Interactions, Mechanism Of Action - DrugBank
-
[PDF] 23.5 BASICITY AND ACIDITY OF AMINES
-
[PDF] MEDCH400_PKA.pdf
-
What Is The PKa Of Methylamine - ECHEMI