Ammonium Cyanide - Wikipedia
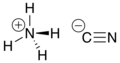 | |
 | |
| Identifiers | |
|---|---|
| CAS Number |
|
| 3D model (JSmol) |
|
| ChemSpider |
|
| PubChem CID |
|
| UNII |
|
| CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | [NH4]CN |
| Molar mass | 44.0559 g/mol |
| Appearance | colourless crystalline solid |
| Density | 1.02 g/cm3 |
| Melting point | 36 °C (decomp.) |
| Solubility in water | very soluble |
| Solubility | very soluble in alcohol |
| Structure | |
| Crystal structure | cubic |
| Related compounds | |
| Other anions |
|
| Other cations |
|
| Related compounds |
|
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Ammonium cyanide is an unstable inorganic compound with the chemical formula NH4CN. It is the ammonium salt of hydrogen cyanide. It consists of ammonium cations NH+4 and cyanide anions CN−. Its structural formula is [NH4]+[C≡N]−.
Uses
[edit]Ammonium cyanide is generally used in organic synthesis.[citation needed] Being unstable, it is not shipped or sold commercially.
Preparation
[edit]Ammonium cyanide is prepared by combining solutions of hydrogen cyanide and ammonia:[citation needed]
HCN + NH3 → NH4CNIt may be prepared by the reaction of calcium cyanide and ammonium carbonate:[citation needed]
Ca(CN)2 + (NH4)2CO3 → 2 NH4CN + CaCO3In dry state, ammonium cyanide is made by heating a mixture of potassium cyanide or potassium ferrocyanide with ammonium chloride and condensing the vapours into ammonium cyanide crystals:[citation needed]
KCN + NH4Cl → NH4CN + KClReactions
[edit]Ammonium cyanide decomposes to ammonia and hydrogen cyanide, often forming a black polymer of hydrogen cyanide:[1]
NH4CN → NH3 + HCNIt undergoes salt metathesis reaction in solution with a number of metal salts to form metal–cyanide complexes.
Reaction with ketones and aldehydes yield aminonitriles, as in the first step of the Strecker amino acid synthesis:
NH4CN + (CH3)2CO → (CH3)2C(NH2)CN + H2OToxicity
[edit] Further information: Cyanide poisoningAmmonium cyanide is highly toxic.
Notes
[edit]- ^ Matthews, Clifford N (1991). "Hydrogen cyanide polymerization: A preferred cosmochemical pathway". Bioastronomy: The Search for Extraterrestrial Life—The Exploration Broadens. Lecture Notes in Physics. Vol. 390. pp. 85–87. doi:10.1007/3-540-54752-5_195. ISBN 978-3-540-54752-5.
References
[edit]- A. F. Wells, Structural Inorganic Chemistry, 5th ed., Oxford University Press, Oxford, UK, 1984.
| |||||||
|---|---|---|---|---|---|---|---|
| Inorganic salts |
| ||||||
| Organic salts |
| ||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Từ khóa » Nh4cn + H2o
-
NH4CN + H2O = HCN + NH4OH - Trình Cân Bằng Phản ứng Hoá Học
-
NH4CN + H2O = NH3 + H3O + CN - Chemical Equation Balancer
-
Acid-base Equilibrium Of NH4CN - Chemistry Stack Exchange
-
CH3COCH3 + NH4CN = H2O + NH2CH2CH2CH2CN | Balanced
-
Hydrogen Cyanide React With Ammonium Hydroxide
-
NH4CN Is A Salt Of Weak Acid HCN (Ka = 6.2 × 10 - Toppr
-
Is An Aqueous Solution Of NH 4 CN Acid Basic Or ... - Course Hero
-
PH Problem : R/chemhelp - Reddit
-
Acid-base Equilibrium Of NH4CN - ECHEMI
-
CH3COCH3 + NH4CN → H2O + NH2CH2CH2CH2CN | Phương ...
-
CN Is A Salt Of Weak Acid HCN(Ka - Tardigrade App
-
When Nh4cn Is Dissolved In Water The Solution Will Be?
-
Solved nsider A Solution Of NH4CN(aq). A. Write The