An Aromatic Compound Of Molecular Formula C6H4Br2 Was Class 12 ...
CoursesCourses for KidsFree study materialOffline CentresMore Store
Store

 Answer
Answer Question Answers for Class 12
Question Answers for Class 12 Class 12 BiologyClass 12 ChemistryClass 12 EnglishClass 12 MathsClass 12 PhysicsClass 12 Social ScienceClass 12 Business StudiesClass 12 EconomicsQuestion Answers for Class 11
Class 12 BiologyClass 12 ChemistryClass 12 EnglishClass 12 MathsClass 12 PhysicsClass 12 Social ScienceClass 12 Business StudiesClass 12 EconomicsQuestion Answers for Class 11 Class 11 EconomicsClass 11 Computer ScienceClass 11 BiologyClass 11 ChemistryClass 11 EnglishClass 11 MathsClass 11 PhysicsClass 11 Social ScienceClass 11 AccountancyClass 11 Business StudiesQuestion Answers for Class 10
Class 11 EconomicsClass 11 Computer ScienceClass 11 BiologyClass 11 ChemistryClass 11 EnglishClass 11 MathsClass 11 PhysicsClass 11 Social ScienceClass 11 AccountancyClass 11 Business StudiesQuestion Answers for Class 10 Class 10 ScienceClass 10 EnglishClass 10 MathsClass 10 Social ScienceClass 10 General KnowledgeQuestion Answers for Class 9
Class 10 ScienceClass 10 EnglishClass 10 MathsClass 10 Social ScienceClass 10 General KnowledgeQuestion Answers for Class 9 Class 9 General KnowledgeClass 9 ScienceClass 9 EnglishClass 9 MathsClass 9 Social ScienceQuestion Answers for Class 8
Class 9 General KnowledgeClass 9 ScienceClass 9 EnglishClass 9 MathsClass 9 Social ScienceQuestion Answers for Class 8 Class 8 ScienceClass 8 EnglishClass 8 MathsClass 8 Social ScienceQuestion Answers for Class 7
Class 8 ScienceClass 8 EnglishClass 8 MathsClass 8 Social ScienceQuestion Answers for Class 7 Class 7 ScienceClass 7 EnglishClass 7 MathsClass 7 Social ScienceQuestion Answers for Class 6
Class 7 ScienceClass 7 EnglishClass 7 MathsClass 7 Social ScienceQuestion Answers for Class 6 Class 6 ScienceClass 6 EnglishClass 6 MathsClass 6 Social ScienceQuestion Answers for Class 5
Class 6 ScienceClass 6 EnglishClass 6 MathsClass 6 Social ScienceQuestion Answers for Class 5 Class 5 ScienceClass 5 EnglishClass 5 MathsClass 5 Social ScienceQuestion Answers for Class 4
Class 5 ScienceClass 5 EnglishClass 5 MathsClass 5 Social ScienceQuestion Answers for Class 4 Class 4 ScienceClass 4 EnglishClass 4 Maths
Class 4 ScienceClass 4 EnglishClass 4 Maths
 An aromatic compound of molecular formula \[C_6H_4Br_2\] was nitrated then only one product of formula \[C_6H_3Br_2NO_2\] was obtained. The original compound is:A.O-dibromobenzeneB.M-dibromobenzeneC.P-dibromobenzeneD.Both A and CAnswer
An aromatic compound of molecular formula \[C_6H_4Br_2\] was nitrated then only one product of formula \[C_6H_3Br_2NO_2\] was obtained. The original compound is:A.O-dibromobenzeneB.M-dibromobenzeneC.P-dibromobenzeneD.Both A and CAnswer Verified580.5k+ viewsHint:Halogens like bromine, are electron withdrawing groups which deactivates the ring, and it also shows inductive effect. -The inductive effect overpowers the resonance effect, so it ultimately becomes ortho para directing, to the electrophilic substitution reaction.Complete answer:-Halogens have the tendency of withdrawing electrons due to high electronegativity and electron affinity. They have a very high inductive effect and electron withdrawing power. In electrophilic substitution reactions it shows both electron donating resonance effect and electron withdrawing inductive effect, but the inductive effect overpowers resonance effect so it deactivates benzene ring and directs it to ortho para positions.-It is given that the reactant should have such a structure, which gives only one compound as a product, which means it should have symmetry in its structure. -The two bromine groups are attached to para position to each other in p-dibromobenzene. Nitration of p-dibromobenzene gives only one product that is \[1,4-dibromo-2-nitrobenzene\], this is because, no matter where the nitro group gets attached it will always give the same compound. In other words the four attacking sites are identical because of the presence of two bromine groups at the para position to each other. This reaction can be represented as,
Verified580.5k+ viewsHint:Halogens like bromine, are electron withdrawing groups which deactivates the ring, and it also shows inductive effect. -The inductive effect overpowers the resonance effect, so it ultimately becomes ortho para directing, to the electrophilic substitution reaction.Complete answer:-Halogens have the tendency of withdrawing electrons due to high electronegativity and electron affinity. They have a very high inductive effect and electron withdrawing power. In electrophilic substitution reactions it shows both electron donating resonance effect and electron withdrawing inductive effect, but the inductive effect overpowers resonance effect so it deactivates benzene ring and directs it to ortho para positions.-It is given that the reactant should have such a structure, which gives only one compound as a product, which means it should have symmetry in its structure. -The two bromine groups are attached to para position to each other in p-dibromobenzene. Nitration of p-dibromobenzene gives only one product that is \[1,4-dibromo-2-nitrobenzene\], this is because, no matter where the nitro group gets attached it will always give the same compound. In other words the four attacking sites are identical because of the presence of two bromine groups at the para position to each other. This reaction can be represented as, 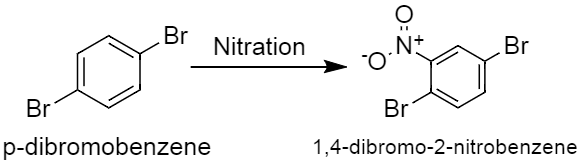 The other option given in the question, where the bromine groups are in meta position to each other, nitration of that compound will at least give two products. One of them will be major and the other one will be a minor product. In case of o-dibromobenzene, the bromine groups are in ortho position to each other, which will also give at least two products, if they undergo the process of nitration. As we can see, the only appropriate option which is correct, is option C, as it gives only one product which matches the description given in the question.So the correct answer is, option C. Note:-Bromine is an electron withdrawing groups, because of presence of seven electrons in its outermost shell-It needs only one electron to complete its octet, and to become stable, that is why it is electronegative in natureRecently Updated PagesBasicity of sulphurous acid and sulphuric acid are
The other option given in the question, where the bromine groups are in meta position to each other, nitration of that compound will at least give two products. One of them will be major and the other one will be a minor product. In case of o-dibromobenzene, the bromine groups are in ortho position to each other, which will also give at least two products, if they undergo the process of nitration. As we can see, the only appropriate option which is correct, is option C, as it gives only one product which matches the description given in the question.So the correct answer is, option C. Note:-Bromine is an electron withdrawing groups, because of presence of seven electrons in its outermost shell-It needs only one electron to complete its octet, and to become stable, that is why it is electronegative in natureRecently Updated PagesBasicity of sulphurous acid and sulphuric acid are Master Class 12 English: Engaging Questions & Answers for Success
Master Class 12 English: Engaging Questions & Answers for Success Master Class 12 Social Science: Engaging Questions & Answers for Success
Master Class 12 Social Science: Engaging Questions & Answers for Success Master Class 12 Maths: Engaging Questions & Answers for Success
Master Class 12 Maths: Engaging Questions & Answers for Success Master Class 12 Economics: Engaging Questions & Answers for Success
Master Class 12 Economics: Engaging Questions & Answers for Success Master Class 12 Physics: Engaging Questions & Answers for Success
Master Class 12 Physics: Engaging Questions & Answers for Success Basicity of sulphurous acid and sulphuric acid are
Basicity of sulphurous acid and sulphuric acid are Master Class 12 English: Engaging Questions & Answers for Success
Master Class 12 English: Engaging Questions & Answers for Success Master Class 12 Social Science: Engaging Questions & Answers for Success
Master Class 12 Social Science: Engaging Questions & Answers for Success Master Class 12 Maths: Engaging Questions & Answers for Success
Master Class 12 Maths: Engaging Questions & Answers for Success Master Class 12 Economics: Engaging Questions & Answers for Success
Master Class 12 Economics: Engaging Questions & Answers for Success Master Class 12 Physics: Engaging Questions & Answers for Success
Master Class 12 Physics: Engaging Questions & Answers for Success
 Giving reasons state the signs positive or negative class 12 physics CBSE
Giving reasons state the signs positive or negative class 12 physics CBSE Explain esterification reaction with the help of a class 12 chemistry CBSE
Explain esterification reaction with the help of a class 12 chemistry CBSE What is defined as a solenoid Depict a diagram with class 12 physics CBSE
What is defined as a solenoid Depict a diagram with class 12 physics CBSE Explain sex determination in humans with line diag class 12 biology CBSE
Explain sex determination in humans with line diag class 12 biology CBSE Organisms of a higher trophic level which feed on several class 12 biology CBSE
Organisms of a higher trophic level which feed on several class 12 biology CBSE Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE
Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE Giving reasons state the signs positive or negative class 12 physics CBSE
Giving reasons state the signs positive or negative class 12 physics CBSE Explain esterification reaction with the help of a class 12 chemistry CBSE
Explain esterification reaction with the help of a class 12 chemistry CBSE What is defined as a solenoid Depict a diagram with class 12 physics CBSE
What is defined as a solenoid Depict a diagram with class 12 physics CBSE Explain sex determination in humans with line diag class 12 biology CBSE
Explain sex determination in humans with line diag class 12 biology CBSE Organisms of a higher trophic level which feed on several class 12 biology CBSE
Organisms of a higher trophic level which feed on several class 12 biology CBSE
Talk to our experts
1800-120-456-456
Sign In- Question Answer
- Class 12
- Chemistry
- An aromatic compound of molecu...


 Question Answers for Class 12
Question Answers for Class 12
 An aromatic compound of molecular formula \[C_6H_4Br_2\] was nitrated then only one product of formula \[C_6H_3Br_2NO_2\] was obtained. The original compound is:A.O-dibromobenzeneB.M-dibromobenzeneC.P-dibromobenzeneD.Both A and CAnswer
An aromatic compound of molecular formula \[C_6H_4Br_2\] was nitrated then only one product of formula \[C_6H_3Br_2NO_2\] was obtained. The original compound is:A.O-dibromobenzeneB.M-dibromobenzeneC.P-dibromobenzeneD.Both A and CAnswer The other option given in the question, where the bromine groups are in meta position to each other, nitration of that compound will at least give two products. One of them will be major and the other one will be a minor product. In case of o-dibromobenzene, the bromine groups are in ortho position to each other, which will also give at least two products, if they undergo the process of nitration. As we can see, the only appropriate option which is correct, is option C, as it gives only one product which matches the description given in the question.So the correct answer is, option C. Note:-Bromine is an electron withdrawing groups, because of presence of seven electrons in its outermost shell-It needs only one electron to complete its octet, and to become stable, that is why it is electronegative in natureRecently Updated PagesBasicity of sulphurous acid and sulphuric acid are
The other option given in the question, where the bromine groups are in meta position to each other, nitration of that compound will at least give two products. One of them will be major and the other one will be a minor product. In case of o-dibromobenzene, the bromine groups are in ortho position to each other, which will also give at least two products, if they undergo the process of nitration. As we can see, the only appropriate option which is correct, is option C, as it gives only one product which matches the description given in the question.So the correct answer is, option C. Note:-Bromine is an electron withdrawing groups, because of presence of seven electrons in its outermost shell-It needs only one electron to complete its octet, and to become stable, that is why it is electronegative in natureRecently Updated PagesBasicity of sulphurous acid and sulphuric acid are Master Class 12 English: Engaging Questions & Answers for Success
Master Class 12 English: Engaging Questions & Answers for Success Master Class 12 Social Science: Engaging Questions & Answers for Success
Master Class 12 Social Science: Engaging Questions & Answers for Success Master Class 12 Maths: Engaging Questions & Answers for Success
Master Class 12 Maths: Engaging Questions & Answers for Success Master Class 12 Economics: Engaging Questions & Answers for Success
Master Class 12 Economics: Engaging Questions & Answers for Success Master Class 12 Physics: Engaging Questions & Answers for Success
Master Class 12 Physics: Engaging Questions & Answers for Success Basicity of sulphurous acid and sulphuric acid are
Basicity of sulphurous acid and sulphuric acid are Master Class 12 English: Engaging Questions & Answers for Success
Master Class 12 English: Engaging Questions & Answers for Success Master Class 12 Social Science: Engaging Questions & Answers for Success
Master Class 12 Social Science: Engaging Questions & Answers for Success Master Class 12 Maths: Engaging Questions & Answers for Success
Master Class 12 Maths: Engaging Questions & Answers for Success Master Class 12 Economics: Engaging Questions & Answers for Success
Master Class 12 Economics: Engaging Questions & Answers for Success Master Class 12 Physics: Engaging Questions & Answers for Success
Master Class 12 Physics: Engaging Questions & Answers for Success
- 1
- 2
 Giving reasons state the signs positive or negative class 12 physics CBSE
Giving reasons state the signs positive or negative class 12 physics CBSE Explain esterification reaction with the help of a class 12 chemistry CBSE
Explain esterification reaction with the help of a class 12 chemistry CBSE What is defined as a solenoid Depict a diagram with class 12 physics CBSE
What is defined as a solenoid Depict a diagram with class 12 physics CBSE Explain sex determination in humans with line diag class 12 biology CBSE
Explain sex determination in humans with line diag class 12 biology CBSE Organisms of a higher trophic level which feed on several class 12 biology CBSE
Organisms of a higher trophic level which feed on several class 12 biology CBSE Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE
Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE Giving reasons state the signs positive or negative class 12 physics CBSE
Giving reasons state the signs positive or negative class 12 physics CBSE Explain esterification reaction with the help of a class 12 chemistry CBSE
Explain esterification reaction with the help of a class 12 chemistry CBSE What is defined as a solenoid Depict a diagram with class 12 physics CBSE
What is defined as a solenoid Depict a diagram with class 12 physics CBSE Explain sex determination in humans with line diag class 12 biology CBSE
Explain sex determination in humans with line diag class 12 biology CBSE Organisms of a higher trophic level which feed on several class 12 biology CBSE
Organisms of a higher trophic level which feed on several class 12 biology CBSE
- 1
- 2
Repeaters Course for NEET 2022 - 23
NEET Repeater 2023 - Aakrosh 1 Year CourseTừ khóa » C6 H4 Br2
-
1,4-Dibromobenzene | C6H4Br2 - PubChem
-
Molar Mass Of (C6H4)Br2
-
C6H4 + Br2 = C6H4Br2 + HBr - Balanced Chemical Equation
-
C6H4(OH)2 + Br2 = C6H4(OH)2Br2 - Phương Trình Hoá Học đã Cân ...
-
An Aromatic Compound Of Molecular Formula C6H4Br2 Was ... - Toppr
-
An Aromatic Compound Of Molecular Formula C6H4Br2 Was ... - Toppr
-
An Aromatic Compound Of Molecular Formula C(6)H(4)Br(2) Was ...
-
Reactions With Aromatic Compounds Of Recoiling Bromine Atoms ...
-
[PDF] An Aqueous Solution Of KBrO3 (550 Mg, 3.3 Mmol) And KBr (800 Mg ...
-
C6 Br2 H4 - CAS号查询 - 爱化学
-
AMAP Chemicals - Arctic Monitoring And Assessment Programme
-
Information Card For Entry 2008030 - Crystallography Open Database
-
Arylation Of [60]Fullerene With Br2/FeCl3/PhH - Taylor & Francis Online