Evaluate The Following: 1.C 4,4 2. C7,3+C11,9 3. C - Gauthmath
Math Resources/Math/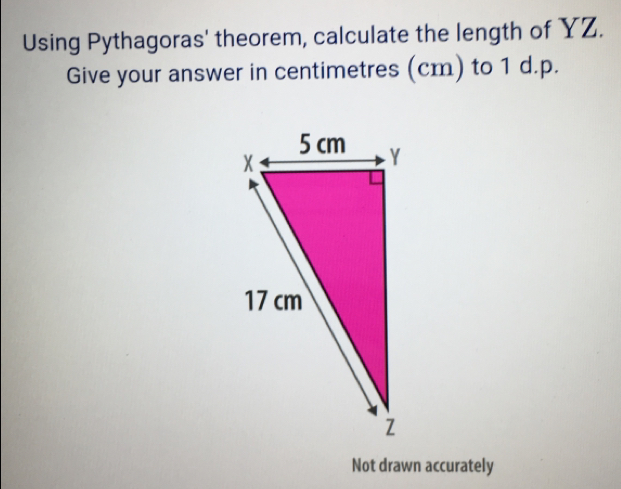 SHOW LESS132
SHOW LESS132![user avatar image]() Answer\overline {YZ} = 2 \sqrt{66} \;cmExplanationDetail stepsPythagorean Theorem: \overline {XY}^{2} + \overline {YZ}^{2} = \overline {XZ}^{2}Substitute \overline {XY} = 5\;cm , \overline {XZ} = 17\;cm into \overline {XY}^{2} + \overline {YZ}^{2} = \overline {XZ}^{2}: 5^{2} + \overline {YZ}^{2} = 17^{2}Calculate 5^{2} + \overline {YZ}^{2} = 17^{2}: \overline {YZ} = 2 \sqrt{66}\;cmClick to rate:99.8(245 votes)Search questionBy textBy image/screenshotDrop your file here orClick Hereto upload
Answer\overline {YZ} = 2 \sqrt{66} \;cmExplanationDetail stepsPythagorean Theorem: \overline {XY}^{2} + \overline {YZ}^{2} = \overline {XZ}^{2}Substitute \overline {XY} = 5\;cm , \overline {XZ} = 17\;cm into \overline {XY}^{2} + \overline {YZ}^{2} = \overline {XZ}^{2}: 5^{2} + \overline {YZ}^{2} = 17^{2}Calculate 5^{2} + \overline {YZ}^{2} = 17^{2}: \overline {YZ} = 2 \sqrt{66}\;cmClick to rate:99.8(245 votes)Search questionBy textBy image/screenshotDrop your file here orClick Hereto upload