Pernicious Anemia: Practice Essentials, Pathophysiology, Etiology
Có thể bạn quan tâm
Cobalamin is an organometallic substance containing a corrin ring, a centrally located cobalt atom, and various axial ligands (see the image below).
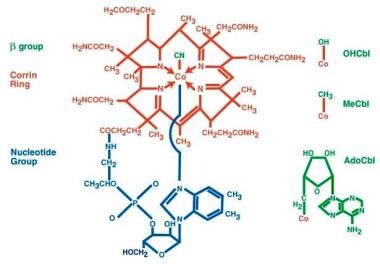 Pernicious anemia. The structure of cyanocobalamin is depicted. The cyanide (Cn) is in green. Other forms of cobalamin (Cbl) include hydroxocobalamin (OHCbl), methylcobalamin (MeCbl), and deoxyadenosylcobalamin (AdoCbl). In these forms, the beta-group is substituted for Cn. The corrin ring with a central cobalt atom is shown in red and the benzimidazole unit in blue. The corrin ring has 4 pyrroles, which bind to the cobalt atom. The fifth substituent is a derivative of dimethylbenzimidazole. The sixth substituent can be Cn, CC3, hydroxycorticosteroid (OH), or deoxyadenosyl. The cobalt atom can be in a +1, +2, or +3 oxidation state. In hydroxocobalamin, it is in the +3 state. The cobalt atom is reduced in a nicotinamide adenine dinucleotide (NADH)–dependent reaction to yield the active coenzyme. It catalyzes 2 types of reactions, which involve either rearrangements (conversion of l methylmalonyl coenzyme A [CoA] to succinyl CoA) or methylation (synthesis of methionine). View Media Gallery
Pernicious anemia. The structure of cyanocobalamin is depicted. The cyanide (Cn) is in green. Other forms of cobalamin (Cbl) include hydroxocobalamin (OHCbl), methylcobalamin (MeCbl), and deoxyadenosylcobalamin (AdoCbl). In these forms, the beta-group is substituted for Cn. The corrin ring with a central cobalt atom is shown in red and the benzimidazole unit in blue. The corrin ring has 4 pyrroles, which bind to the cobalt atom. The fifth substituent is a derivative of dimethylbenzimidazole. The sixth substituent can be Cn, CC3, hydroxycorticosteroid (OH), or deoxyadenosyl. The cobalt atom can be in a +1, +2, or +3 oxidation state. In hydroxocobalamin, it is in the +3 state. The cobalt atom is reduced in a nicotinamide adenine dinucleotide (NADH)–dependent reaction to yield the active coenzyme. It catalyzes 2 types of reactions, which involve either rearrangements (conversion of l methylmalonyl coenzyme A [CoA] to succinyl CoA) or methylation (synthesis of methionine). View Media Gallery The basic structure known as vitamin B12 is solely synthesized by microorganisms, but most animals are capable of converting vitamin B12 into the two coenzyme forms, adenosylcobalamin and methylcobalamin. The former is required for conversion of L-methylmalonic acid to succinyl coenzyme A (CoA), and the latter acts as a methyltransferase for conversion of homocysteine to methionine.
When either cobalamin or folate is deficient, thymidine synthase function is impaired. This leads to megaloblastic changes in all rapidly dividing cells because DNA synthesis is diminished. In erythroid precursors, macrocytosis and ineffective erythropoiesis occur.
Severe neurologic impairment, usually subacute combined system degeneration, occurs in cobalamin deficiency. However, vitamin B12 deficiency can also present as peripheral neuropathy, psychosis, or leukoencephalopathy. Neurologic manifestations may occur independently of hematologic manifestations in pernicious anemia. The biochemical impairment in neurologic degeneration may differ from hematologic changes. [3]
Meat and milk are the main dietary sources of cobalamin. Because body stores of cobalamin usually exceed 1000 µg and the daily requirement is about 1 µg, strict adherence to a vegetarian diet for more than 5 years usually is required to produce findings of cobalamin deficiency.
Dietary cobalamin is absorbed in a series of steps, which require proteolytic release from foodstuffs and binding to IF. Subsequently, recognition of the IF-cobalamin complex by specialized ileal receptors—cubilin receptors—must occur for transport into the portal circulation, where it is bound by transcobalamin II (TCII), which serves as the plasma transporter.
The cobalamin-TCII complex binds to cell surfaces and is endocytosed. The transcobalamin is degraded within a lysozyme, and the cobalamin is released into the cytoplasm. An enzyme-mediated reduction of the cobalt occurs with either cytoplasmic methylation to form methylcobalamin or mitochondrial adenosylation to form adenosylcobalamin.
Defects of these steps produce manifestations of cobalamin dysfunction. Most defects become manifest in infancy and early childhood and result in impaired development, intellecual disability, and a macrocytic anemia. Certain defects cause methylmalonic aciduria and homocystinuria. See the image below.
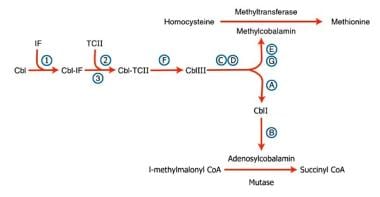 Pernicious anemia. Inherited disorders of cobalamin (Cbl) metabolism are depicted. The numbers and letters correspond to the sites at which abnormalities have been identified, as follows: (1) absence of intrinsic factor (IF); (2) abnormal Cbl intestinal adsorption; and (3) abnormal transcobalamin II (TC II), (a) mitochondrial Cbl reduction (Cbl A), (b) cobalamin adenosyl transferase (Cbl B), (c and d) cytosolic Cbl metabolism (Cbl C and D), (e and g) methyl transferase Cbl utilization (Cbl E and G), and (f) lysosomal Cbl efflux (Cbl F). View Media Gallery
Pernicious anemia. Inherited disorders of cobalamin (Cbl) metabolism are depicted. The numbers and letters correspond to the sites at which abnormalities have been identified, as follows: (1) absence of intrinsic factor (IF); (2) abnormal Cbl intestinal adsorption; and (3) abnormal transcobalamin II (TC II), (a) mitochondrial Cbl reduction (Cbl A), (b) cobalamin adenosyl transferase (Cbl B), (c and d) cytosolic Cbl metabolism (Cbl C and D), (e and g) methyl transferase Cbl utilization (Cbl E and G), and (f) lysosomal Cbl efflux (Cbl F). View Media Gallery Intrinsic factor is a gastric protein secreted by parietal cells that is necessary for vitamin B12 absorption. Pernicious anemia is an autoimmune disorder that leads to insufficient intrinsic factor levels either as a result of auto-antibody mediated destruction of parietal cells and/or the intrinsic factor protein itself. Parietal cell auto-antibodies target gastric H+/K+-ATPase. [4] Other disorders that interfere with the absorption and metabolism of vitamin B12 can also result in cobalamin deficiency, with the development of a macrocytic anemia and neurologic complications.
Antiparietal cell antibodies occur in 90% of patients with pernicious anemia but in only 5% of healthy adults. Similarly, binding and blocking antibodies to IF are found in most patients with pernicious anemia. A greater association than anticipated exists between pernicious anemia and other autoimmune diseases, including thyroid disorders, type 1 diabetes mellitus, ulcerative colitis, Addison disease, infertility, and acquired agammaglobulinemia.
An association between pernicious anemia and Helicobacter pylori infections has been postulated but not clearly proven. [5] A higher prevalence of H pylori infection has been reported in patients with pernicious anemia, and eradication of H pylori has had potentially therapeutic effects (eg, reductions in antiparietal cell antibodies, amelioration of atrophic gastritis. However, further research on this hypothesis is warranted. [6]
Cobalamin deficiency may result from dietary insufficiency of vitamin B12; disorders of the stomach, small bowel, and pancreas; certain infections; and abnormalities of transport, metabolism, and utilization (see Etiology). Deficiency may be observed in strict vegetarians. [7] Breastfed infants of vegetarian mothers also are affected. Severely affected infants of vegetarian mothers who do not have overt cobalamin deficiency have been reported. [8]
Classic pernicious anemia produces cobalamin deficiency due to failure of the stomach to secrete IF (see the image below).
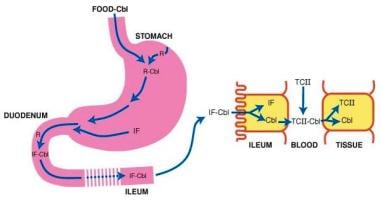 Pernicious anemia. Cobalamin (Cbl) is freed from meat in the acidic milieu of the stomach where it binds R factors in competition with intrinsic factor (IF). Cbl is freed from R factors in the duodenum by proteolytic digestion of the R factors by pancreatic enzymes. The IF-Cbl complex transits to the ileum where it is bound to ileal receptors. The IF-Cbl enters the ileal absorptive cell, and the Cbl is released and enters the plasma. In the plasma, the Cbl is bound to transcobalamin II (TC II), which delivers the complex to nonintestinal cells. In these cells, Cbl is freed from the transport protein. View Media Gallery
Pernicious anemia. Cobalamin (Cbl) is freed from meat in the acidic milieu of the stomach where it binds R factors in competition with intrinsic factor (IF). Cbl is freed from R factors in the duodenum by proteolytic digestion of the R factors by pancreatic enzymes. The IF-Cbl complex transits to the ileum where it is bound to ileal receptors. The IF-Cbl enters the ileal absorptive cell, and the Cbl is released and enters the plasma. In the plasma, the Cbl is bound to transcobalamin II (TC II), which delivers the complex to nonintestinal cells. In these cells, Cbl is freed from the transport protein. View Media Gallery In adults, pernicious anemia is associated with severe gastric atrophy and achlorhydria, which are irreversible. The achlorhydria results in a decrease in the release of cobalamin bound to dietary protein. [1] Coexistent iron deficiency is common because achlorhydria prevents solubilization of dietary ferric iron from food. Autoimmune phenomena and thyroid disease frequently are observed. Patients with pernicious anemia have a 2- to 3-fold increased incidence of gastric carcinoma.
Từ khóa » Vit B12 Deficiency Anemia Name
-
Vitamin B12 Or Folate Deficiency Anaemia - Causes - NHS
-
Pernicious Anemia - Wikipedia
-
Vitamin B12 Deficiency Anemia | Johns Hopkins Medicine
-
Pernicious Anemia: Symptoms, Causes, And Treatments - Healthline
-
Vitamin B12–Deficiency Anemia | NHLBI, NIH
-
Vitamin B12 Deficiency - StatPearls - NCBI Bookshelf
-
Pernicious Anemia: MedlinePlus Medical Encyclopedia
-
Vitamin B12 Deficiency Anemia: MedlinePlus Medical Encyclopedia
-
Pernicious Anemia: Definition, Symptoms, Causes & Treatment
-
Vitamin B12 Deficiency Anemia: Symptoms, Causes, Treatments
-
Other Names For Pernicious Anemia
-
Vitamin B12 Deficiency: Causes, Symptoms, And Treatment - WebMD
-
Vitamin B12 Deficiency - Nutritional Disorders - MSD Manuals
-
Vitamin Deficiency Anemia - Symptoms And Causes - Mayo Clinic