SOLVED:i Need Help For I J And K. 2 CoCl2 + 2 NH4Cl + H2O2 + 10 ...

Question
i need help for I J and K. 2 CoCl2 + 2 NH4Cl + H2O2 + 10 NH3 ----> 2 Co(NH3)6Cl3 + 2 H2O 1) You are assigned the following amount of CoCl2. Volume of CoCl2 assigned and used 4.94 mL Concentration of CoCl2 1.79 M To this is added... Mass of charcoal 0.20 g Volume of 15.0 M ammonia 5.0 mL Mass of ammonium chloride 1.42 g The ammonium chloride was dissolved. The mixture was cooled. To this was added: Volume of 10% (by weight) hydrogen peroxide 4.9 mL (density = 1.00 g/mL) Calculate: (Base theoretical values on millimoles CoCl2 used) a) Millimoles of CoCl2 used ___8.8426____________ mmol b) Millimoles of NH3 theoretically required __________44.213_____ mmol c) Millimoles of NH3 actually added __________75_____ mmol d) Millimoles of NH4Cl theoretically required ________8.8426_______ mmol e) Millimoles of NH4Cl actually added ___________26.546____ mmol f) Millimoles of H2O2 theoretically required ___________4.4213____ mmol g) Millimoles of H2O2 actually added __________14.40_____ mmol h) Which of the above is the limiting reagent? __________cobalt_____ i) Theoretical yield of Co(NH3)6Cl3 _______________ g After reaction, workup, and recrystallization, the dry product was put into a sample tube, labeled, and turned in. Mass of the empty sample tube 9.4918 g Mass of sample tube with Co(NH3)6Cl3 11.7599 g Calculate: j) Weight of product turned in _____________ g k) The per cent yield of complex _____________ % i need help for I J and K. 2 CoCl2 + 2 NH4Cl + H2O2 + 10 NH3 ----> 2 Co(NH3)6Cl3 + 2 H2O 1) You are assigned the following amount of CoCl2. Volume of CoCl2 assigned and used 4.94 mL Concentration of CoCl2 1.79 M To this is added... Mass of charcoal 0.20 g Volume of 15.0 M ammonia 5.0 mL Mass of ammonium chloride 1.42 g The ammonium chloride was dissolved. The mixture was cooled. To this was added: Volume of 10% (by weight) hydrogen peroxide 4.9 mL (density = 1.00 g/mL) Calculate: (Base theoretical values on millimoles CoCl2 used) a) Millimoles of CoCl2 used ___8.8426____________ mmol b) Millimoles of NH3 theoretically required __________44.213_____ mmol c) Millimoles of NH3 actually added __________75_____ mmol d) Millimoles of NH4Cl theoretically required ________8.8426_______ mmol e) Millimoles of NH4Cl actually added ___________26.546____ mmol f) Millimoles of H2O2 theoretically required ___________4.4213____ mmol g) Millimoles of H2O2 actually added __________14.40_____ mmol h) Which of the above is the limiting reagent? __________cobalt_____ i) Theoretical yield of Co(NH3)6Cl3 _______________ g After reaction, workup, and recrystallization, the dry product was put into a sample tube, labeled, and turned in. Mass of the empty sample tube 9.4918 g Mass of sample tube with Co(NH3)6Cl3 11.7599 g Calculate: j) Weight of product turned in _____________ g k) The per cent yield of complex _____________ % Show more…Added by Ruth O.
Step 1
94 mL) x (1.79 mmol/mL) = 8.8426 mmol Show more…
Show all steps


Please give Ace some feedback
Your feedback will help us improve your experience
 i need help for I J and K. 2 CoCl2 + 2 NH4Cl + H2O2 + 10 NH3 ----> 2 Co(NH3)6Cl3 + 2 H2O 1) You are assigned the following amount of CoCl2. Volume of CoCl2 assigned and used 4.94 mL Concentration of CoCl2 1.79 M To this is added... Mass of charcoal 0.20 g Volume of 15.0 M ammonia 5.0 mL Mass of ammonium chloride 1.42 g The ammonium chloride was dissolved. The mixture was cooled. To this was added: Volume of 10% (by weight) hydrogen peroxide 4.9 mL (density = 1.00 g/mL) Calculate: (Base theoretical values on millimoles CoCl2 used) a) Millimoles of CoCl2 used ___8.8426____________ mmol b) Millimoles of NH3 theoretically required __________44.213_____ mmol c) Millimoles of NH3 actually added __________75_____ mmol d) Millimoles of NH4Cl theoretically required ________8.8426_______ mmol e) Millimoles of NH4Cl actually added ___________26.546____ mmol f) Millimoles of H2O2 theoretically required ___________4.4213____ mmol g) Millimoles of H2O2 actually added __________14.40_____ mmol h) Which of the above is the limiting reagent? __________cobalt_____ i) Theoretical yield of Co(NH3)6Cl3 _______________ g After reaction, workup, and recrystallization, the dry product was put into a sample tube, labeled, and turned in. Mass of the empty sample tube 9.4918 g Mass of sample tube with Co(NH3)6Cl3 11.7599 g Calculate: j) Weight of product turned in _____________ g k) The per cent yield of complex _____________ %
i need help for I J and K. 2 CoCl2 + 2 NH4Cl + H2O2 + 10 NH3 ----> 2 Co(NH3)6Cl3 + 2 H2O 1) You are assigned the following amount of CoCl2. Volume of CoCl2 assigned and used 4.94 mL Concentration of CoCl2 1.79 M To this is added... Mass of charcoal 0.20 g Volume of 15.0 M ammonia 5.0 mL Mass of ammonium chloride 1.42 g The ammonium chloride was dissolved. The mixture was cooled. To this was added: Volume of 10% (by weight) hydrogen peroxide 4.9 mL (density = 1.00 g/mL) Calculate: (Base theoretical values on millimoles CoCl2 used) a) Millimoles of CoCl2 used ___8.8426____________ mmol b) Millimoles of NH3 theoretically required __________44.213_____ mmol c) Millimoles of NH3 actually added __________75_____ mmol d) Millimoles of NH4Cl theoretically required ________8.8426_______ mmol e) Millimoles of NH4Cl actually added ___________26.546____ mmol f) Millimoles of H2O2 theoretically required ___________4.4213____ mmol g) Millimoles of H2O2 actually added __________14.40_____ mmol h) Which of the above is the limiting reagent? __________cobalt_____ i) Theoretical yield of Co(NH3)6Cl3 _______________ g After reaction, workup, and recrystallization, the dry product was put into a sample tube, labeled, and turned in. Mass of the empty sample tube 9.4918 g Mass of sample tube with Co(NH3)6Cl3 11.7599 g Calculate: j) Weight of product turned in _____________ g k) The per cent yield of complex _____________ %  Powered by NumerAI
Powered by NumerAI 


Lizabeth Tumminello and 92 other Chemistry 101 educators are ready to help you.
Ask a new question
Labs
Want to see this concept in action?
NEWExplore this concept interactively to see how it behaves as you change inputs.
View LabsKey Concepts

Recommended Videos

Describe the preparation of the ammonium carbonate/ammonia mixture. Describe what you observed upon combining this solution with the cobalt solution. Write a reaction along the lines given in the text to show the expected cobalt-containing species present in the solution at this point. Procedure for Carbonatotetraamminecobalt (III) Nitrate, [Co(NH3)4CO3]NO3: Dissolve 20 g (0.21 mol) of (NH4)2CO3 in 60 mL of H2O and add 60 mL of concentrated aqueous NH3 (do not worry if all the (NH4)2CO3 does not dissolve in the water as it will dissolve when the ammonium hydroxide is added). While stirring, pour this solution into a solution containing 15 g (0.052 mol) of [Co(OH2)6](NO3)2 in 30 mL of H2O. Then slowly add 8 mL of a 30% H2O2 solution (the 8 mL should be added to the cobalt/ammonia solution 1 mL at a time with stirring - there is a delayed release of the gas which can cause the solution to foam over if the hydrogen peroxide is added all at once). Pour the solution into a large beaker and place it on a hot plate underneath a fume cap. Heat the solution to evaporate the solvent until only ~100 mL is left - the solution temperature may be raised to the 80-85°C range without the risk of decomposing the cobalt-ammine complex you are trying to isolate. However, above 90°C and particularly near the boiling point, decomposition may occur.
Adi S.

Describe the preparation of the ammonium carbonate/ammonia mixture. Describe what you observed upon combining this solution with the cobalt solution. Write a reaction along the lines given in the text to show the expected cobalt-containing species present in the solution at this point. Procedure for Carbonatotetraamminecobalt (III) Nitrate, [Co(NH3)4CO3]NO3: Dissolve 20 g (0.21 mol) of (NH4)2CO3 in 60 mL of H2O and add 60 mL of concentrated aqueous NH3 (do not worry if all the (NH4)2CO3 does not dissolve in the water as it will dissolve when the ammonium hydroxide is added). While stirring, pour this solution into a solution containing 15 g (0.052 mol) of [Co(OH2)6](NO3)2 in 30 mL of H2O. Then slowly add 8 mL of a 30% H2O2 solution (the 8 mL should be added to the cobalt/ammonia solution 1 mL at a time with stirring - there is a delayed release of the gas which can cause the solution to foam over if the hydrogen peroxide is added all at once). Pour the solution into a large beaker and place it on a hot plate underneath a fume cap. Heat the solution to evaporate the solvent until only ~100 mL is left - the solution temperature may be raised to the 80-85°C range without the risk of decomposing the cobalt-ammine complex you are trying to isolate. However, above 90°C and particularly near the boiling point, decomposition may occur.
Adi S.
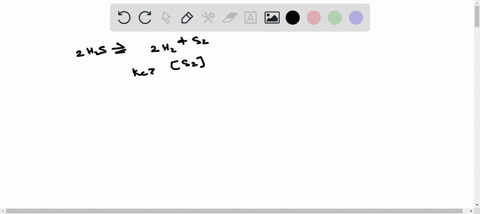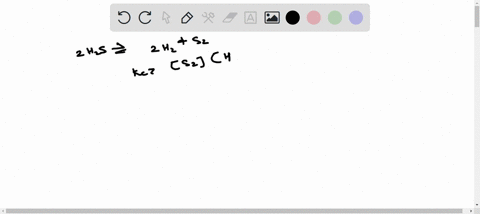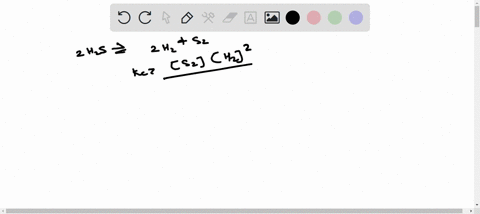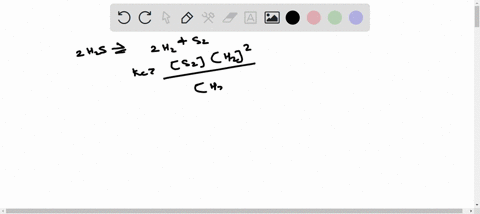
ICE TABLE QUESTIONS Uses significant figures and units effectively. 1. At 225 K, the equilibrium constant for the following reaction is 18.5. 2H2S(g) ⇌ 2H2(g) + S2(g) At equilibrium, the hydrogen sulfide gas concentration was found to be 1.18 mol/L, and the sulfur gas concentration was found to be 1.86 mol/L. a) Determine the equilibrium concentration of the hydrogen gas. b) Determine the initial concentration of the hydrogen sulfide. 2. An initial amount of 1.25 mol of hydrogen gas reacts with 1.25 mol of chlorine gas in a 10.00 L container to form gaseous hydrochloric acid. If the reaction proceeds to equilibrium, determine the equilibrium amounts for all compounds in the reaction. (Keq = 5.00) 3. A 1.0 L container is filled with air. When the container is heated to 1500 K, the nitrogen gas and the oxygen gas in the air react to form nitrous oxide. Initially, the container contained 0.80 mol of nitrogen gas and 0.20 mol of oxygen gas. The equilibrium constant for the reaction is Keq = 1.1 × 10^(-5). Determine the equilibrium concentration of nitrous oxide. 4. At 300 K, the equilibrium constant is Keq = 35.5 for the following reaction: C(s) + H2(g) ⇌ CH4(g) + C2H2(g) The following concentrations are found to be present in a 1.00 L container at a particular point in the process. nC = 3.195 mol [H2] = 0.17 mol/L [CH4] = 1.065 mol/L [C2H2] = 0.885 mol/L Determine if the reaction is at equilibrium. If it is not at equilibrium, determine the direction in which the reaction is favored. 5. The reaction of gaseous carbon dioxide and hydrogen gas to form gaseous carbon monoxide and water vapor has an equilibrium constant of 0.74 at 600 K. If the reaction reaches equilibrium in a 1.50 L container and initially 1.41 mol of carbon dioxide and 2.61 mol of hydrogen gas are reacted, what will the equilibrium concentrations be of the products and reactants?
Dinesh S.
Recommended Textbooks

Chemistry: Structure and Properties
Nivaldo Tro 2nd Edition
Chemistry The Central Science
Theodore L. Brown 14th Edition
Chemistry
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste 10th EditionWhat our educators say
 25992 Students Helped in Chemistry 101 “Numerade has a great goal - to increase people's educational levels all around the world. Educators do not complete student's personal homework tasks. We create video tutorials that may be used for many years in the future.”
25992 Students Helped in Chemistry 101 “Numerade has a great goal - to increase people's educational levels all around the world. Educators do not complete student's personal homework tasks. We create video tutorials that may be used for many years in the future.” 

 44601 Students Helped in Chemistry 101 "The format has forced me to think about what knowledge is needed by the student to solve a problem and present it concisely and understandably within the time constraint of the video."
44601 Students Helped in Chemistry 101 "The format has forced me to think about what knowledge is needed by the student to solve a problem and present it concisely and understandably within the time constraint of the video." 

 30417 Students Helped in Chemistry 101 “Explaining topics while I make Numerade videos has helped me deepen my own understanding and come up with new ways to help my students grasp concepts while I'm teaching.”
30417 Students Helped in Chemistry 101 “Explaining topics while I make Numerade videos has helped me deepen my own understanding and come up with new ways to help my students grasp concepts while I'm teaching.” 

 A free answerjust for you
A free answerjust for you Watch the video solution with this free unlock.
View the Answer

Log in to watch this video ...and 100,000,000 more!
PASSWORD
Log in OR Continue with Facebook Continue with Apple Continue with Clever Don't have an account? Sign UpTừ khóa » Cocl2+nh4cl+nh3+h2o2
-
2 CoCl2 + 2 NH4Cl + H2O2 + 10 NH3 → 2 [Co(NH3)6]Cl3 + 2 H2O
-
CoCl2 + NH4Cl + NH3 + H2O2 = (Co(NH3)5Cl)Cl2 + ... - ChemicalAid
-
CoCl2 + NH4Cl + NH3 + H2O2 = (Co(NH3)6)Cl3 + H2O - ChemicalAid
-
CHM361 LABORATORY REPORT Exp cx - Course Hero
-
[PDF] CHEM. 1CL LAB FINALFriday June 7, 4-5 PM* Rooms To Be ...
-
Solved CoCl2 + NH4Cl + NH3 + H2O2 → [Co(NH3)5Cl]Cl2 + - Chegg
-
Write Chemical Equations For The Following Reactions : A) CoCl2 + ...
-
Chemistry/Experiments - Science Made Alive
-
Chem Lab 2: Lab 3 Flashcards - Quizlet
-
Consider: 2 CoCl2(aq) + 2 NH4Cl(aq) + 8 NH3(aq) + H2O2(aq)
-
Hello, I Am Trying To Write A Chemical Equation For A Reaction I Did And ...
-
[PDF] Metallic Pigments Introduction Coloured Compounds Are Used In A ...
-
Cobalt(II) Chloride React With Ammonia, Ammonium ...