The Reaction Of Ethanol With Conc. H2SO4gives - Vedantu
Có thể bạn quan tâm
CoursesCourses for KidsFree study materialOffline CentresMore Store
Store

 Answer
Answer Question Answers for Class 12
Question Answers for Class 12 Class 12 BiologyClass 12 ChemistryClass 12 EnglishClass 12 MathsClass 12 PhysicsClass 12 Social ScienceClass 12 Business StudiesClass 12 EconomicsQuestion Answers for Class 11
Class 12 BiologyClass 12 ChemistryClass 12 EnglishClass 12 MathsClass 12 PhysicsClass 12 Social ScienceClass 12 Business StudiesClass 12 EconomicsQuestion Answers for Class 11 Class 11 EconomicsClass 11 Computer ScienceClass 11 BiologyClass 11 ChemistryClass 11 EnglishClass 11 MathsClass 11 PhysicsClass 11 Social ScienceClass 11 AccountancyClass 11 Business StudiesQuestion Answers for Class 10
Class 11 EconomicsClass 11 Computer ScienceClass 11 BiologyClass 11 ChemistryClass 11 EnglishClass 11 MathsClass 11 PhysicsClass 11 Social ScienceClass 11 AccountancyClass 11 Business StudiesQuestion Answers for Class 10 Class 10 ScienceClass 10 EnglishClass 10 MathsClass 10 Social ScienceClass 10 General KnowledgeQuestion Answers for Class 9
Class 10 ScienceClass 10 EnglishClass 10 MathsClass 10 Social ScienceClass 10 General KnowledgeQuestion Answers for Class 9 Class 9 General KnowledgeClass 9 ScienceClass 9 EnglishClass 9 MathsClass 9 Social ScienceQuestion Answers for Class 8
Class 9 General KnowledgeClass 9 ScienceClass 9 EnglishClass 9 MathsClass 9 Social ScienceQuestion Answers for Class 8 Class 8 ScienceClass 8 EnglishClass 8 MathsClass 8 Social ScienceQuestion Answers for Class 7
Class 8 ScienceClass 8 EnglishClass 8 MathsClass 8 Social ScienceQuestion Answers for Class 7 Class 7 ScienceClass 7 EnglishClass 7 MathsClass 7 Social ScienceQuestion Answers for Class 6
Class 7 ScienceClass 7 EnglishClass 7 MathsClass 7 Social ScienceQuestion Answers for Class 6 Class 6 ScienceClass 6 EnglishClass 6 MathsClass 6 Social ScienceQuestion Answers for Class 5
Class 6 ScienceClass 6 EnglishClass 6 MathsClass 6 Social ScienceQuestion Answers for Class 5 Class 5 ScienceClass 5 EnglishClass 5 MathsClass 5 Social ScienceQuestion Answers for Class 4
Class 5 ScienceClass 5 EnglishClass 5 MathsClass 5 Social ScienceQuestion Answers for Class 4 Class 4 ScienceClass 4 EnglishClass 4 Maths
Class 4 ScienceClass 4 EnglishClass 4 Maths
 The reaction of ethanol with conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$gives: A. ethaneB. etheneC. ethyneD. ethanoic acid Answer
The reaction of ethanol with conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$gives: A. ethaneB. etheneC. ethyneD. ethanoic acid Answer Verified589.5k+ viewsHint: The reaction between ethanol and conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ is a dehydration reaction. Dehydration means removal of water molecules to form alkenes. As concentrated ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ is a dehydrating agent. Ethanol has two carbon atoms and an alcohol group and rest vacancies are filled by hydrogen atoms. Complete answer: Let us form the product of this reaction using the mechanism:Step (1)- Attack of ${{\text{H}}^{+}}$ ion on the lone pair of oxygen atom, as the ${{\text{H}}^{+}}$ ion is electron deficient and lone pairs are electron rich. The ${{\text{H}}^{+}}$ ion is formed from dissociation of sulphuric acid into its respective ions $\left( \text{2}{{\text{H}}^{+}}+\text{SO}_{4}^{2-} \right)$. This attack on lone pairs leads to formation of protonated alcohol.
Verified589.5k+ viewsHint: The reaction between ethanol and conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ is a dehydration reaction. Dehydration means removal of water molecules to form alkenes. As concentrated ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ is a dehydrating agent. Ethanol has two carbon atoms and an alcohol group and rest vacancies are filled by hydrogen atoms. Complete answer: Let us form the product of this reaction using the mechanism:Step (1)- Attack of ${{\text{H}}^{+}}$ ion on the lone pair of oxygen atom, as the ${{\text{H}}^{+}}$ ion is electron deficient and lone pairs are electron rich. The ${{\text{H}}^{+}}$ ion is formed from dissociation of sulphuric acid into its respective ions $\left( \text{2}{{\text{H}}^{+}}+\text{SO}_{4}^{2-} \right)$. This attack on lone pairs leads to formation of protonated alcohol. 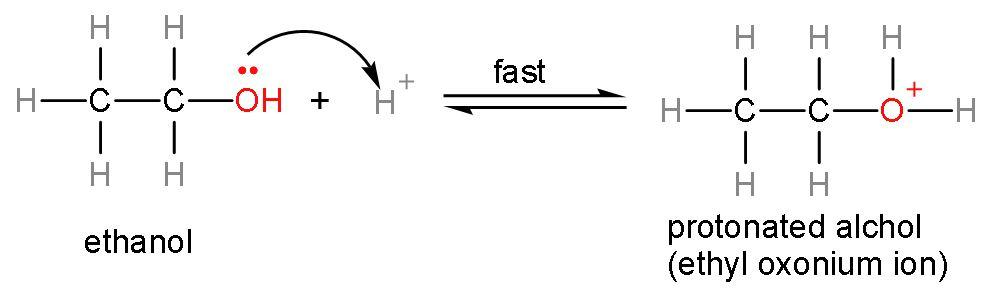 Step (2)- Removal of water occurs from the protonated alcohol to form carbocation. This is a slow step or rate-determining step. The rate of any reaction is determined by the slowest step.
Step (2)- Removal of water occurs from the protonated alcohol to form carbocation. This is a slow step or rate-determining step. The rate of any reaction is determined by the slowest step. 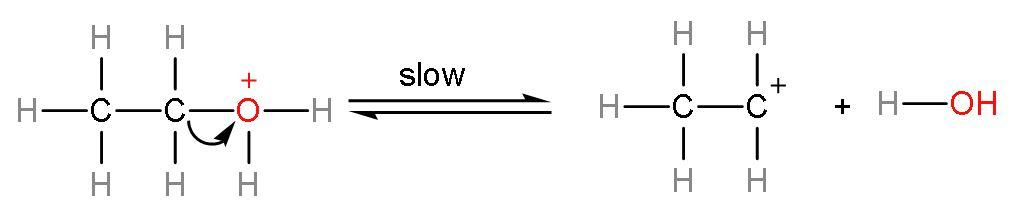 Step (3)- Formation of alkene or ethene takes place by the elimination of ${{\text{H}}^{+}}$ion from the carbocation. The ${{\text{H}}^{+}}$ion is a catalyst of this reaction as it remains unutilized.
Step (3)- Formation of alkene or ethene takes place by the elimination of ${{\text{H}}^{+}}$ion from the carbocation. The ${{\text{H}}^{+}}$ion is a catalyst of this reaction as it remains unutilized. 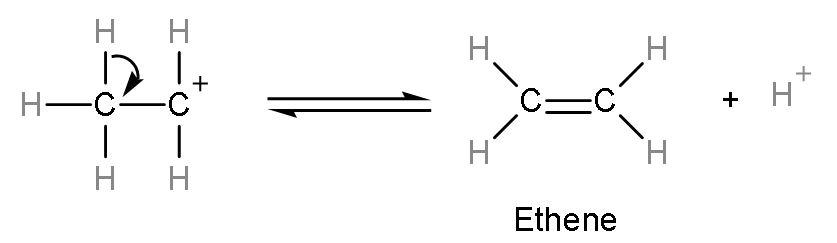 The correct answer to this question is ethene is formed as a product on reaction between ethanol and conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ So, the correct answer is “Option B”.Additional Information:Secondary and tertiary alcohols dehydrate under mild conditions only because after protonation, the carbocation formed is highly stable due to hyperconjugation. Thus, the relative ease of dehydration of alcohols is $\text{Tertiary}>\text{Secondary}>\text{Primary}$. Note: Alcohols reacts with conc.${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ not only to form alkenes but at different temperatures, the reactions are different thus, the products formed are different. Like, (1) At 383 K, ethanol reacts with sulphuric acid to form ethyl hydrogen sulphate and water. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{383\text{K}}{{\text{C}}_{2}}{{\text{H}}_{5}}\text{HS}{{\text{O}}_{4}}+{{\text{H}}_{2}}\text{O}$ (2) At 413 K, ethanol reacts with sulphuric acid to form diethyl ether. As, the two molecules of ethanol combine to remove water. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{413\text{K}}{{\text{C}}_{2}}{{\text{H}}_{5}}{\mathrm O}{{\text{C}}_{2}}{{\text{H}}_{5}}+{{\eta }_{2}}{\mathrm O}$ (3) At 443 K, ethanol reacts with sulphuric acid to form ethene. Ethanol gets protonated and water gets removed. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{443\text{K}}{{\text{C}}_{2}}{{\text{H}}_{4}}+{{\eta }_{2}}{\mathrm O}$ So, the product formed depends on the reagents and as well as on the temperatures.Recently Updated PagesMaster Class 12 English: Engaging Questions & Answers for Success
The correct answer to this question is ethene is formed as a product on reaction between ethanol and conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ So, the correct answer is “Option B”.Additional Information:Secondary and tertiary alcohols dehydrate under mild conditions only because after protonation, the carbocation formed is highly stable due to hyperconjugation. Thus, the relative ease of dehydration of alcohols is $\text{Tertiary}>\text{Secondary}>\text{Primary}$. Note: Alcohols reacts with conc.${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ not only to form alkenes but at different temperatures, the reactions are different thus, the products formed are different. Like, (1) At 383 K, ethanol reacts with sulphuric acid to form ethyl hydrogen sulphate and water. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{383\text{K}}{{\text{C}}_{2}}{{\text{H}}_{5}}\text{HS}{{\text{O}}_{4}}+{{\text{H}}_{2}}\text{O}$ (2) At 413 K, ethanol reacts with sulphuric acid to form diethyl ether. As, the two molecules of ethanol combine to remove water. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{413\text{K}}{{\text{C}}_{2}}{{\text{H}}_{5}}{\mathrm O}{{\text{C}}_{2}}{{\text{H}}_{5}}+{{\eta }_{2}}{\mathrm O}$ (3) At 443 K, ethanol reacts with sulphuric acid to form ethene. Ethanol gets protonated and water gets removed. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{443\text{K}}{{\text{C}}_{2}}{{\text{H}}_{4}}+{{\eta }_{2}}{\mathrm O}$ So, the product formed depends on the reagents and as well as on the temperatures.Recently Updated PagesMaster Class 12 English: Engaging Questions & Answers for Success Master Class 12 Social Science: Engaging Questions & Answers for Success
Master Class 12 Social Science: Engaging Questions & Answers for Success Master Class 12 Maths: Engaging Questions & Answers for Success
Master Class 12 Maths: Engaging Questions & Answers for Success Master Class 12 Economics: Engaging Questions & Answers for Success
Master Class 12 Economics: Engaging Questions & Answers for Success Master Class 12 Physics: Engaging Questions & Answers for Success
Master Class 12 Physics: Engaging Questions & Answers for Success Master Class 12 Business Studies: Engaging Questions & Answers for Success
Master Class 12 Business Studies: Engaging Questions & Answers for Success Master Class 12 English: Engaging Questions & Answers for Success
Master Class 12 English: Engaging Questions & Answers for Success Master Class 12 Social Science: Engaging Questions & Answers for Success
Master Class 12 Social Science: Engaging Questions & Answers for Success Master Class 12 Maths: Engaging Questions & Answers for Success
Master Class 12 Maths: Engaging Questions & Answers for Success Master Class 12 Economics: Engaging Questions & Answers for Success
Master Class 12 Economics: Engaging Questions & Answers for Success Master Class 12 Physics: Engaging Questions & Answers for Success
Master Class 12 Physics: Engaging Questions & Answers for Success Master Class 12 Business Studies: Engaging Questions & Answers for Success
Master Class 12 Business Studies: Engaging Questions & Answers for Success
 Draw the diagram showing the germination of pollen class 12 biology CBSE
Draw the diagram showing the germination of pollen class 12 biology CBSE Differentiate between insitu conservation and exsitu class 12 biology CBSE
Differentiate between insitu conservation and exsitu class 12 biology CBSE RNA and DNA are chiral molecules their chirality is class 12 chemistry CBSE
RNA and DNA are chiral molecules their chirality is class 12 chemistry CBSE Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE
Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE What is Saheb looking for in the garbage dump Where class 12 english CBSE
What is Saheb looking for in the garbage dump Where class 12 english CBSE What are the major means of transport Explain each class 12 social science CBSE
What are the major means of transport Explain each class 12 social science CBSE Draw the diagram showing the germination of pollen class 12 biology CBSE
Draw the diagram showing the germination of pollen class 12 biology CBSE Differentiate between insitu conservation and exsitu class 12 biology CBSE
Differentiate between insitu conservation and exsitu class 12 biology CBSE RNA and DNA are chiral molecules their chirality is class 12 chemistry CBSE
RNA and DNA are chiral molecules their chirality is class 12 chemistry CBSE Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE
Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE What is Saheb looking for in the garbage dump Where class 12 english CBSE
What is Saheb looking for in the garbage dump Where class 12 english CBSE
Talk to our experts
1800-120-456-456
Sign In- Question Answer
- Class 12
- Chemistry
- The reaction of ethanol with c...


 Question Answers for Class 12
Question Answers for Class 12
 The reaction of ethanol with conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$gives: A. ethaneB. etheneC. ethyneD. ethanoic acid Answer
The reaction of ethanol with conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$gives: A. ethaneB. etheneC. ethyneD. ethanoic acid Answer Step (2)- Removal of water occurs from the protonated alcohol to form carbocation. This is a slow step or rate-determining step. The rate of any reaction is determined by the slowest step.
Step (2)- Removal of water occurs from the protonated alcohol to form carbocation. This is a slow step or rate-determining step. The rate of any reaction is determined by the slowest step.  Step (3)- Formation of alkene or ethene takes place by the elimination of ${{\text{H}}^{+}}$ion from the carbocation. The ${{\text{H}}^{+}}$ion is a catalyst of this reaction as it remains unutilized.
Step (3)- Formation of alkene or ethene takes place by the elimination of ${{\text{H}}^{+}}$ion from the carbocation. The ${{\text{H}}^{+}}$ion is a catalyst of this reaction as it remains unutilized.  The correct answer to this question is ethene is formed as a product on reaction between ethanol and conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ So, the correct answer is “Option B”.Additional Information:Secondary and tertiary alcohols dehydrate under mild conditions only because after protonation, the carbocation formed is highly stable due to hyperconjugation. Thus, the relative ease of dehydration of alcohols is $\text{Tertiary}>\text{Secondary}>\text{Primary}$. Note: Alcohols reacts with conc.${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ not only to form alkenes but at different temperatures, the reactions are different thus, the products formed are different. Like, (1) At 383 K, ethanol reacts with sulphuric acid to form ethyl hydrogen sulphate and water. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{383\text{K}}{{\text{C}}_{2}}{{\text{H}}_{5}}\text{HS}{{\text{O}}_{4}}+{{\text{H}}_{2}}\text{O}$ (2) At 413 K, ethanol reacts with sulphuric acid to form diethyl ether. As, the two molecules of ethanol combine to remove water. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{413\text{K}}{{\text{C}}_{2}}{{\text{H}}_{5}}{\mathrm O}{{\text{C}}_{2}}{{\text{H}}_{5}}+{{\eta }_{2}}{\mathrm O}$ (3) At 443 K, ethanol reacts with sulphuric acid to form ethene. Ethanol gets protonated and water gets removed. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{443\text{K}}{{\text{C}}_{2}}{{\text{H}}_{4}}+{{\eta }_{2}}{\mathrm O}$ So, the product formed depends on the reagents and as well as on the temperatures.Recently Updated PagesMaster Class 12 English: Engaging Questions & Answers for Success
The correct answer to this question is ethene is formed as a product on reaction between ethanol and conc. ${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ So, the correct answer is “Option B”.Additional Information:Secondary and tertiary alcohols dehydrate under mild conditions only because after protonation, the carbocation formed is highly stable due to hyperconjugation. Thus, the relative ease of dehydration of alcohols is $\text{Tertiary}>\text{Secondary}>\text{Primary}$. Note: Alcohols reacts with conc.${{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}$ not only to form alkenes but at different temperatures, the reactions are different thus, the products formed are different. Like, (1) At 383 K, ethanol reacts with sulphuric acid to form ethyl hydrogen sulphate and water. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{383\text{K}}{{\text{C}}_{2}}{{\text{H}}_{5}}\text{HS}{{\text{O}}_{4}}+{{\text{H}}_{2}}\text{O}$ (2) At 413 K, ethanol reacts with sulphuric acid to form diethyl ether. As, the two molecules of ethanol combine to remove water. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{413\text{K}}{{\text{C}}_{2}}{{\text{H}}_{5}}{\mathrm O}{{\text{C}}_{2}}{{\text{H}}_{5}}+{{\eta }_{2}}{\mathrm O}$ (3) At 443 K, ethanol reacts with sulphuric acid to form ethene. Ethanol gets protonated and water gets removed. ${{\text{C}}_{2}}{{\text{H}}_{5}}\text{OH}+\text{conc}\text{.}{{\text{H}}_{2}}\text{S}{{\text{O}}_{4}}\xrightarrow{443\text{K}}{{\text{C}}_{2}}{{\text{H}}_{4}}+{{\eta }_{2}}{\mathrm O}$ So, the product formed depends on the reagents and as well as on the temperatures.Recently Updated PagesMaster Class 12 English: Engaging Questions & Answers for Success Master Class 12 Social Science: Engaging Questions & Answers for Success
Master Class 12 Social Science: Engaging Questions & Answers for Success Master Class 12 Maths: Engaging Questions & Answers for Success
Master Class 12 Maths: Engaging Questions & Answers for Success Master Class 12 Economics: Engaging Questions & Answers for Success
Master Class 12 Economics: Engaging Questions & Answers for Success Master Class 12 Physics: Engaging Questions & Answers for Success
Master Class 12 Physics: Engaging Questions & Answers for Success Master Class 12 Business Studies: Engaging Questions & Answers for Success
Master Class 12 Business Studies: Engaging Questions & Answers for Success Master Class 12 English: Engaging Questions & Answers for Success
Master Class 12 English: Engaging Questions & Answers for Success Master Class 12 Social Science: Engaging Questions & Answers for Success
Master Class 12 Social Science: Engaging Questions & Answers for Success Master Class 12 Maths: Engaging Questions & Answers for Success
Master Class 12 Maths: Engaging Questions & Answers for Success Master Class 12 Economics: Engaging Questions & Answers for Success
Master Class 12 Economics: Engaging Questions & Answers for Success Master Class 12 Physics: Engaging Questions & Answers for Success
Master Class 12 Physics: Engaging Questions & Answers for Success Master Class 12 Business Studies: Engaging Questions & Answers for Success
Master Class 12 Business Studies: Engaging Questions & Answers for Success
- 1
- 2
 Draw the diagram showing the germination of pollen class 12 biology CBSE
Draw the diagram showing the germination of pollen class 12 biology CBSE Differentiate between insitu conservation and exsitu class 12 biology CBSE
Differentiate between insitu conservation and exsitu class 12 biology CBSE RNA and DNA are chiral molecules their chirality is class 12 chemistry CBSE
RNA and DNA are chiral molecules their chirality is class 12 chemistry CBSE Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE
Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE What is Saheb looking for in the garbage dump Where class 12 english CBSE
What is Saheb looking for in the garbage dump Where class 12 english CBSE What are the major means of transport Explain each class 12 social science CBSE
What are the major means of transport Explain each class 12 social science CBSE Draw the diagram showing the germination of pollen class 12 biology CBSE
Draw the diagram showing the germination of pollen class 12 biology CBSE Differentiate between insitu conservation and exsitu class 12 biology CBSE
Differentiate between insitu conservation and exsitu class 12 biology CBSE RNA and DNA are chiral molecules their chirality is class 12 chemistry CBSE
RNA and DNA are chiral molecules their chirality is class 12 chemistry CBSE Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE
Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE What is Saheb looking for in the garbage dump Where class 12 english CBSE
What is Saheb looking for in the garbage dump Where class 12 english CBSE
- 1
- 2
Repeaters Course for NEET 2022 - 23
NEET Repeater 2023 - Aakrosh 1 Year CourseTừ khóa » Ch3ch2oh H2so4 443k
-
Ethanol Is Heated With Excess Concentrated H2SO4 At 443K.i ... - Toppr
-
What Happens When Ethanol Is Heated At 443 K With Excess ... - Toppr
-
What Happens When Ehanol Is Heated With Concentrated H2so4 At ...
-
What Happens When Ethanol Is Heated At 443 K ... - Sarthaks EConnect
-
Ethene Is Formed When Ethanol At 443k Is Heated With An Excess Of ...
-
On Heating Ethanol With Excess Of Conc. H2SO4 At 443 K ... - Doubtnut
-
Ethene Is Formed When Ethanol At 443 K Is Heated With Excess Of ...
-
What Will Happen When Ethanol Is Heated With Concentrated ... - Quora
-
Dehydrating Alcohols To Make Alkenes - Chemistry LibreTexts
-
When Ethyl Alcohol Is Heated With Conc. H2SO4 At 443 K Ethylene Is ...
-
[Solved] On Heating Ethanol With Concentrated H2SO4 At 443 K Gives
-
Write The Mechanism Of The Reaction Of Ch3ch2oh With Concentrated ...
-
Reaction Of Alcohols With Conc H2SO4 - YouTube
-
Write Chemical Equations To Show What Happens When - Tutorix