When Ammonium (NH4+) Is Placed In Water, A Chemical Reaction ...

Question
When ammonium (NH4+) is placed in water, a chemical reaction occurs and an equilibrium is established as shown below. NH4+ (aq) + H2O (l) ⇄ NH3 (aq) + H3O+ (aq) The pKa of NH4+ is 9.25. Predict whether the acid form (NH4+) or the base form (NH3) is predominant at pH = 7.4. Explain your answer. When ammonium (NH4+) is placed in water, a chemical reaction occurs and an equilibrium is established as shown below. NH4+ (aq) + H2O (l) ⇄ NH3 (aq) + H3O+ (aq) The pKa of NH4+ is 9.25. Predict whether the acid form (NH4+) or the base form (NH3) is predominant at pH = 7.4. Explain your answer. Show more…Added by Kimberly A.
Step 1
The pKa value tells us about the acidity of a substance: the lower the pKa, the stronger the acid. In this case, the pKa of NH4+ is 9.25. This means that NH4+ is a weak acid, as it has a relatively high pKa value. Show more…
Show all steps


Please give Ace some feedback
Your feedback will help us improve your experience
 When ammonium (NH4+) is placed in water, a chemical reaction occurs and an equilibrium is established as shown below. NH4+ (aq) + H2O (l) ⇄ NH3 (aq) + H3O+ (aq) The pKa of NH4+ is 9.25. Predict whether the acid form (NH4+) or the base form (NH3) is predominant at pH = 7.4. Explain your answer.
When ammonium (NH4+) is placed in water, a chemical reaction occurs and an equilibrium is established as shown below. NH4+ (aq) + H2O (l) ⇄ NH3 (aq) + H3O+ (aq) The pKa of NH4+ is 9.25. Predict whether the acid form (NH4+) or the base form (NH3) is predominant at pH = 7.4. Explain your answer.  Powered by NumerAI
Powered by NumerAI 


Alkendra Singh and 80 other Chemistry 101 educators are ready to help you.
Ask a new question
Labs
Want to see this concept in action?
NEWExplore this concept interactively to see how it behaves as you change inputs.
View LabsKey Concepts

Recommended Videos
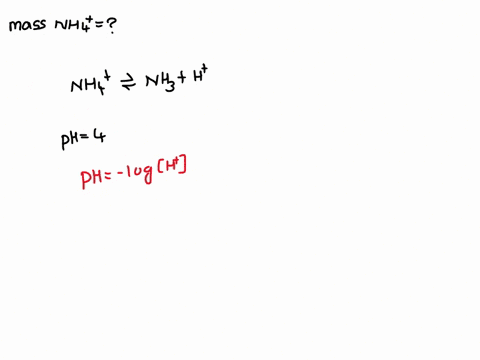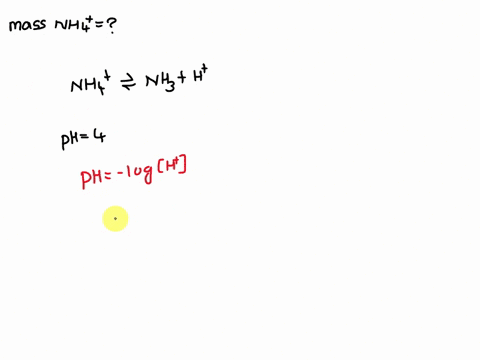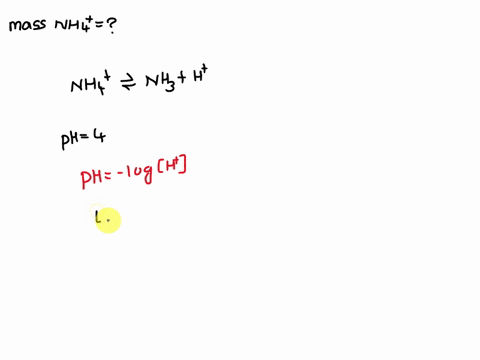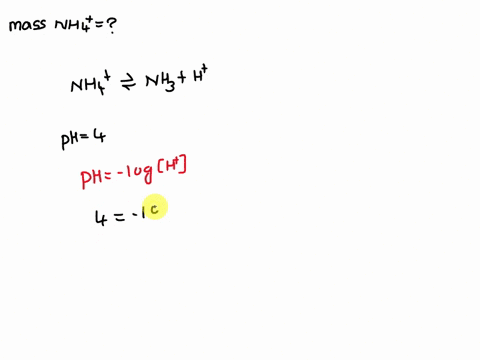
Ammonium (NH4+) is a weak acid with a pKa of 9.26. The dissociation is: NH4+ → NH3 + H+. Calculate the mass of ammonium added to 1 L of water in order to obtain a pH of 4 at equilibrium.
Supreeta N.
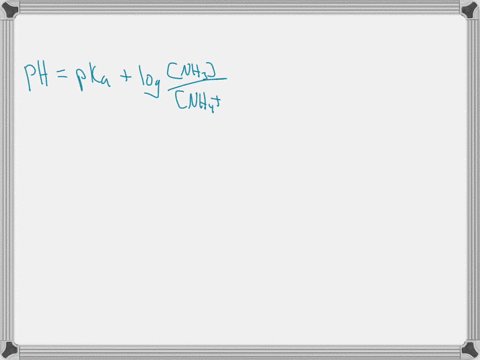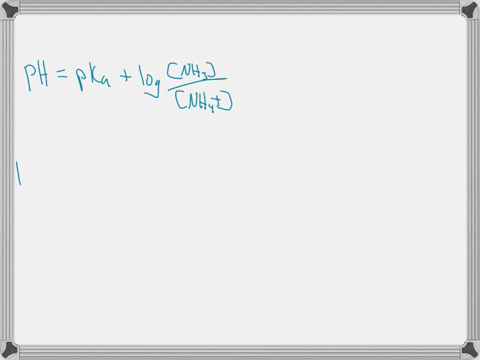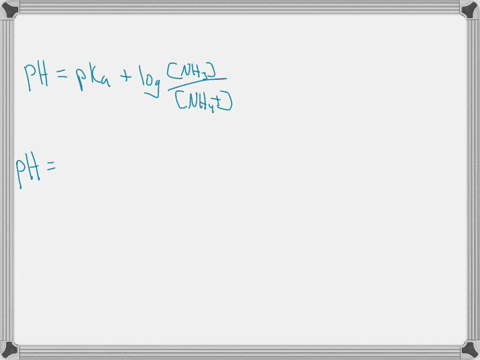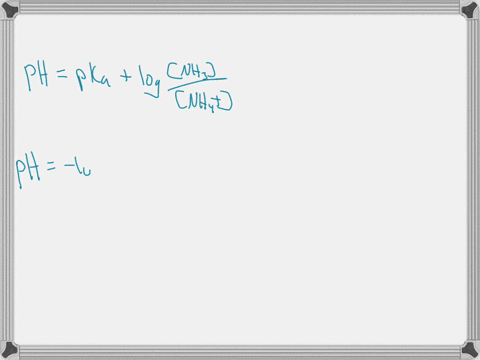
Ammonia (NH3) ionizes according to the following reaction: NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH−(aq) The base dissociation constant for ammonia (NH3) is Kb = 1.8 × 10−5. Ammonia (NH3) also has a chloride salt, ammonium chloride (NH4Cl), which is soluble in water. If 0.070 M of ammonia (NH3) and 0.035 M of its salt ammonium chloride (NH4Cl) are mixed in a solution, what is the pH of this solution? 9.6 4.4 1.3 12.7
David C.
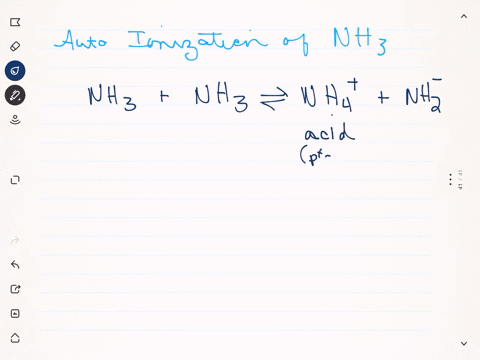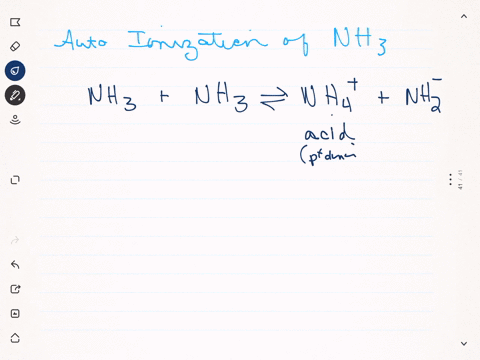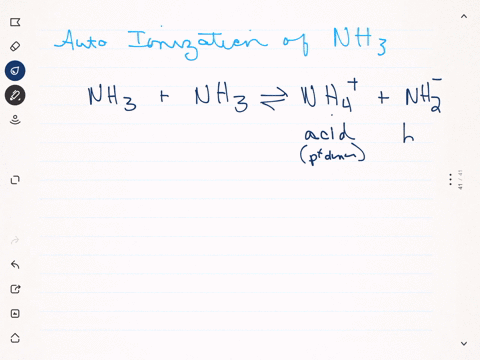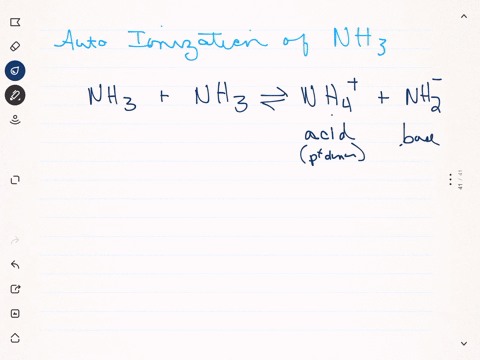
Like water, liquid ammonia undergoes autoionization: $$\mathrm{NH}_{3}+\mathrm{NH}_{3} \rightleftharpoons \mathrm{NH}_{4}^{+}+\mathrm{NH}_{2}^{-}$$ (a) Identify the Bronsted acids and Bronsted bases in this reaction. (b) What species correspond to $\mathrm{H}^{+}$ and $\mathrm{OH}^{-}$ and what is the condition for a neutral solution?

Recommended Textbooks

Chemistry: Structure and Properties
Nivaldo Tro 2nd Edition
Chemistry The Central Science
Theodore L. Brown 14th Edition
Chemistry
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste 10th EditionTranscript
What our educators say
 25992 Students Helped in Chemistry 101 “Numerade has a great goal - to increase people's educational levels all around the world. Educators do not complete student's personal homework tasks. We create video tutorials that may be used for many years in the future.”
25992 Students Helped in Chemistry 101 “Numerade has a great goal - to increase people's educational levels all around the world. Educators do not complete student's personal homework tasks. We create video tutorials that may be used for many years in the future.” 

 44601 Students Helped in Chemistry 101 "The format has forced me to think about what knowledge is needed by the student to solve a problem and present it concisely and understandably within the time constraint of the video."
44601 Students Helped in Chemistry 101 "The format has forced me to think about what knowledge is needed by the student to solve a problem and present it concisely and understandably within the time constraint of the video." 

 30417 Students Helped in Chemistry 101 “Explaining topics while I make Numerade videos has helped me deepen my own understanding and come up with new ways to help my students grasp concepts while I'm teaching.”
30417 Students Helped in Chemistry 101 “Explaining topics while I make Numerade videos has helped me deepen my own understanding and come up with new ways to help my students grasp concepts while I'm teaching.” 

 A free answerjust for you
A free answerjust for you Watch the video solution with this free unlock.
View the Answer

Log in to watch this video ...and 100,000,000 more!
PASSWORD
Log in OR Continue with Facebook Continue with Apple Continue with Clever Don't have an account? Sign UpTừ khóa » Nh4 Plus H20
-
Is This Reaction Right, NH4+H2O=H3O+NH3? - Quora
-
NH4{+} + H2O = NH3 + H3O{+} - Chemical Equation Balancer
-
NH3 + H2O → OH:- + NH4:+ - Chemical Equations Online!
-
The Reaction Of Water With Ammonia Is H2O + NH3→ NH4^+ + OH
-
The Acid & Base Components Of Ammonium Chloride - Sciencing
-
Hydrolysis Of Salts
-
Conjugate Acid And Conjugate Base For The Reaction NH3 + H2O
-
Equation For NH4Cl + H2O (Ammonium Chloride + Water) - YouTube
-
How To Balance NH3 + H2O = NH4OH (ammonia And Water)
-
Solved In The Following Reaction: NH4+ + H2O = NH3 + H3O+ A
-
Ammonium Sulfate | (NH4)2SO4 - PubChem
-
Gas Phase Solvation Of The Ammonium Ion By NH3 And H2O And ...
-
What Is The Chemical Reaction NH3 H2O NH4 OH?