2.12: Electronegativity - Chemistry LibreTexts
Maybe your like
Electronegativity
The elements with the highest ionization energies are generally those with the most negative electron affinities, which are located toward the upper right corner of the periodic table (compare Figure 2.9.2 and Figure 2.10.2). Conversely, the elements with the lowest ionization energies are generally those with the least negative electron affinities and are located in the lower left corner of the periodic table.
Because the tendency of an element to gain or lose electrons is so important in determining its chemistry, various methods have been developed to quantitatively describe this tendency. The most important method uses a measurement called electronegativity (represented by the Greek letter chi, χ, pronounced “ky” as in “sky”), defined as the relative ability of an atom to attract electrons to itself in a chemical compound. Elements with high electronegativities tend to acquire electrons in chemical reactions and are found in the upper right corner of the periodic table. Elements with low electronegativities tend to lose electrons in chemical reactions and are found in the lower left corner of the periodic table.
Unlike ionization energy or electron affinity, the electronegativity of an atom is not a simple, fixed property that can be directly measured in a single experiment. In fact, an atom’s electronegativity should depend to some extent on its chemical environment because the properties of an atom are influenced by its neighbors in a chemical compound. Nevertheless, when different methods for measuring the electronegativity of an atom are compared, they all tend to assign similar relative values to a given element. For example, all scales predict that fluorine has the highest electronegativity and cesium the lowest of the stable elements, which suggests that all the methods are measuring the same fundamental property.
Note
Electronegativity is defined as the ability of an atom in a particular molecule to attract electrons to itself. The greater the value, the greater the attractiveness for electrons.
Electronegativity is a function of: (1) the atom's ionization energy (how strongly the atom holds on to its own electrons) and (2) the atom's electron affinity (how strongly the atom attracts other electrons). Both of these are properties of the isolated atom. An element that is will be highly electronegative has:
- a large (negative) electron affinity
- a high ionization energy (always endothermic, or positive for neutral atoms)
and will
- attract electrons from other atoms
- resist having its own electrons attracted away.
The Pauling Electronegativity Scale
The original electronegativity scale, developed in the 1930s by Linus Pauling (1901– 1994) was based on measurements of the strengths of covalent bonds between different elements. Pauling arbitrarily set the electronegativity of fluorine at 4.0 (although today it has been refined to 3.98), thereby creating a scale in which all elements have values between 0 and 4.0.
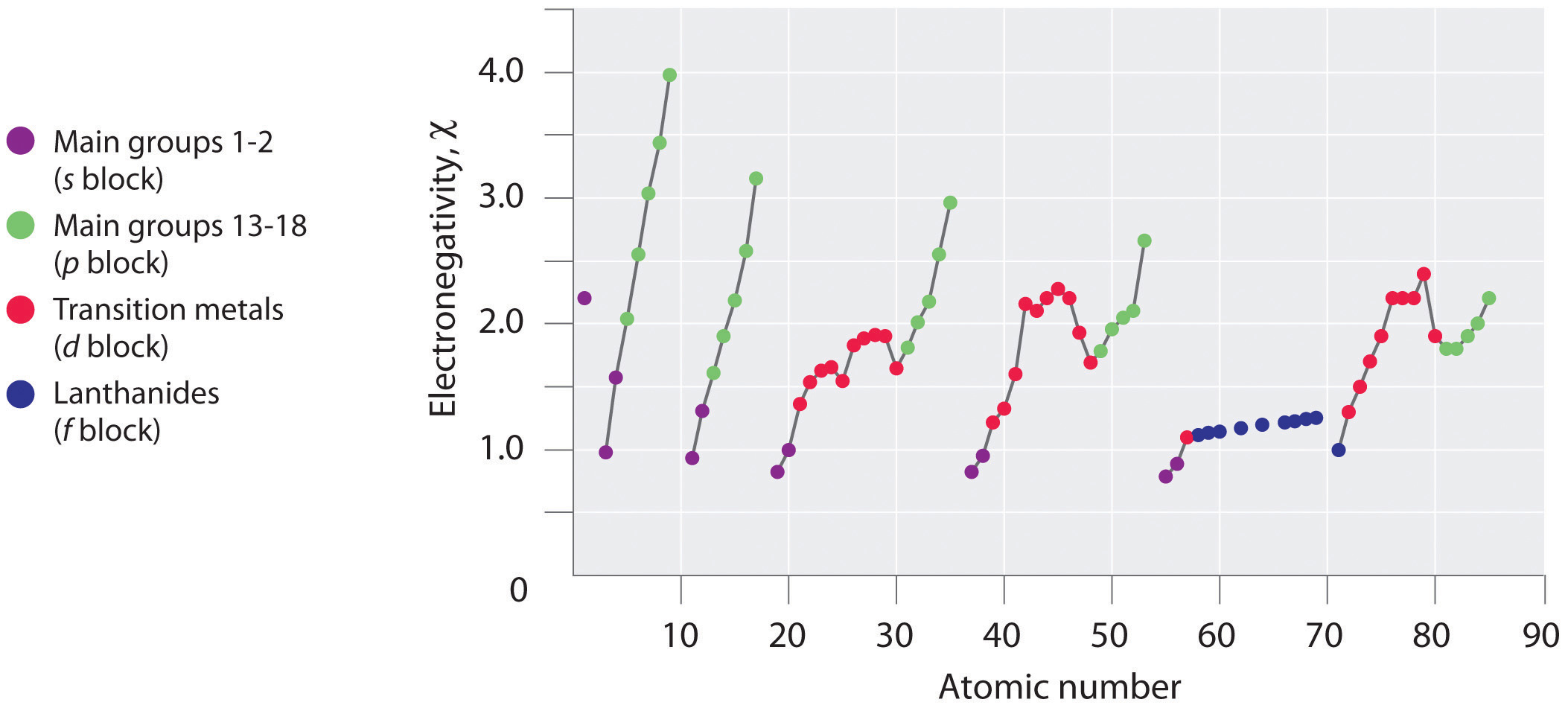
Periodic variations in Pauling’s electronegativity values are illustrated in Figure 2.12.1 and Figure 2.12.2. If we ignore the inert gases and elements for which no stable isotopes are known, we see that fluorine (\(\chi = 3.98\)) is the most electronegative element and cesium is the least electronegative nonradioactive element (\(\chi = 0.79\)). Because electronegativities generally increase diagonally from the lower left to the upper right of the periodic table, elements lying on diagonal lines running from upper left to lower right tend to have comparable values (e.g., O and Cl and N, S, and Br).
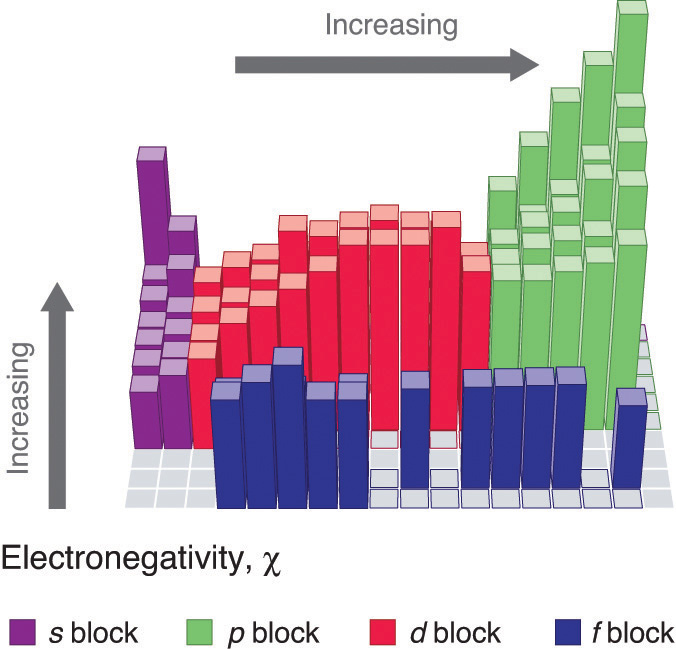
Figure 2.12.2: Pauling Electronegativity Values of the s-, p-, d-, and f-Block Elements. Values for most of the actinides are approximate. Elements for which no data are available are shown in gray. Source: Data from L. Pauling, The Nature of the Chemical Bond, 3rd ed. (1960).
Linus Pauling (1901-1994)
Pauling won two Nobel Prizes, one for chemistry in 1954 and one for peace in 1962. When he was nine, Pauling’s father died, and his mother tried to convince him to quit school to support the family. He did not quit school but was denied a high school degree because of his refusal to take a civics class.
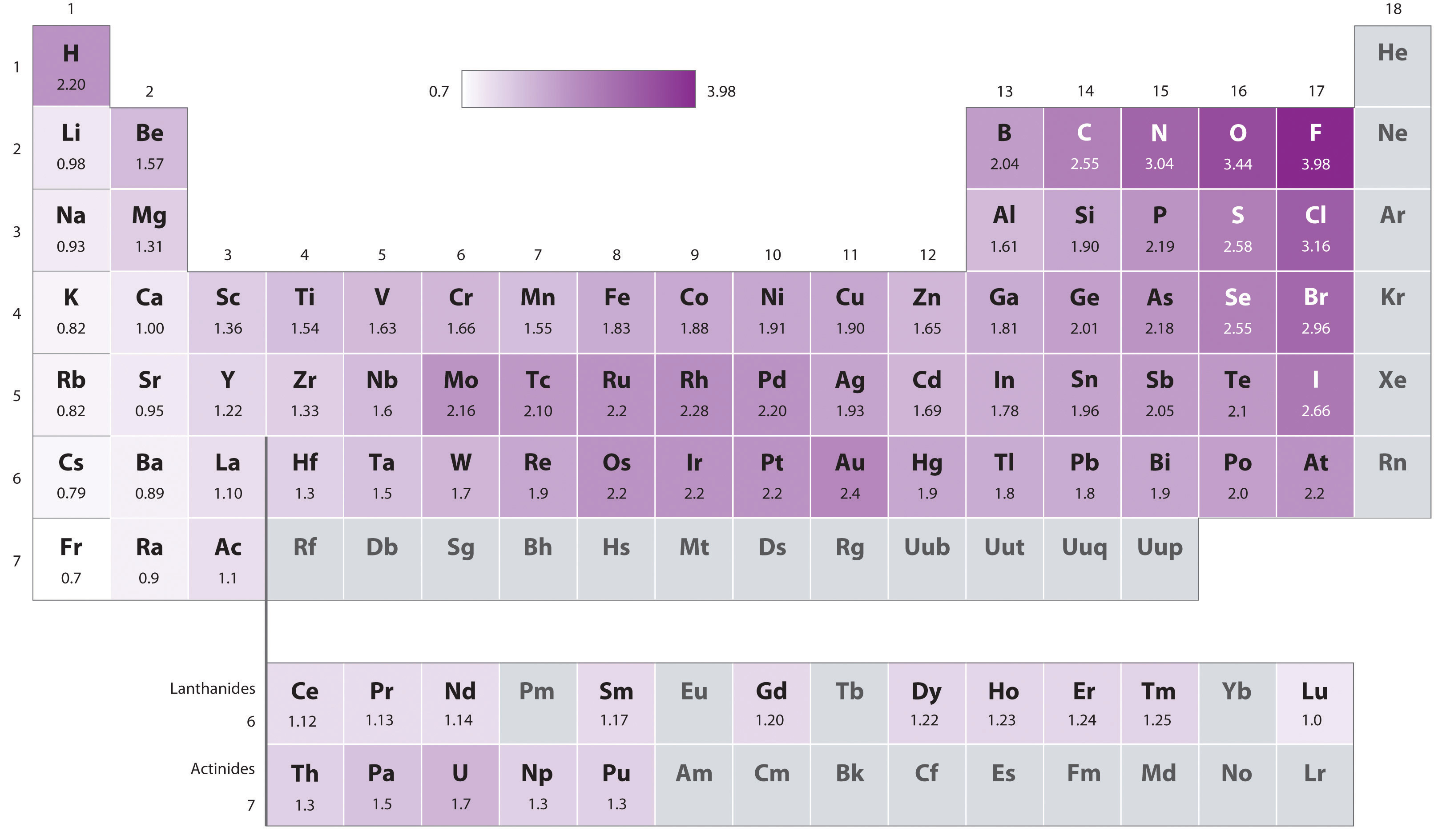
Pauling’s method is limited by the fact that many elements do not form stable covalent compounds with other elements; hence their electronegativities cannot be measured by his method. Other definitions have since been developed that address this problem (e.g., the Mulliken electronegativity scale).
Tag » A Reactive Element With A Relatively High Electronegativity
-
Solved QUESTION 10 A Reactive Element With A Relatively High
-
A Reactive Element With A Relatively High ...
-
Chemistry Chapter 7 Review Flashcards | Quizlet
-
A Reactive Element With A Relatively High Electronegativity Would Be ...
-
QUESTION 10 A Reactive Element With A Relatively High … - Study QA
-
23.9: Halogens- Reactive Chemicals With High Electronegativity
-
A Reactive Element With A Relatively High Electronegativity Would ...
-
Which Of The Following Elements Have Relatively High ... - Toppr
-
[PDF] 4/4/2015 CHEM 177- Chapter 7 Review TINA AKINYI
-
A Al 2 O 3 B MgO C NaCl D AlF 3 2 8 A Reactive Element With A ...
-
Electronegativity Seen As The Ground-State Average Valence ...
-
Periodic Table - Model Science Software