23.9: Halogens- Reactive Chemicals With High Electronegativity
Maybe your like
Reactions and Compounds of the Noble Gases
For many years, it was thought that the only compounds the noble gases could form were clathrates. Clathrates are solid compounds in which a gas, the guest, occupies holes in a lattice formed by a less volatile, chemically dissimilar substance, the host (Figure \(\PageIndex{1}\)).
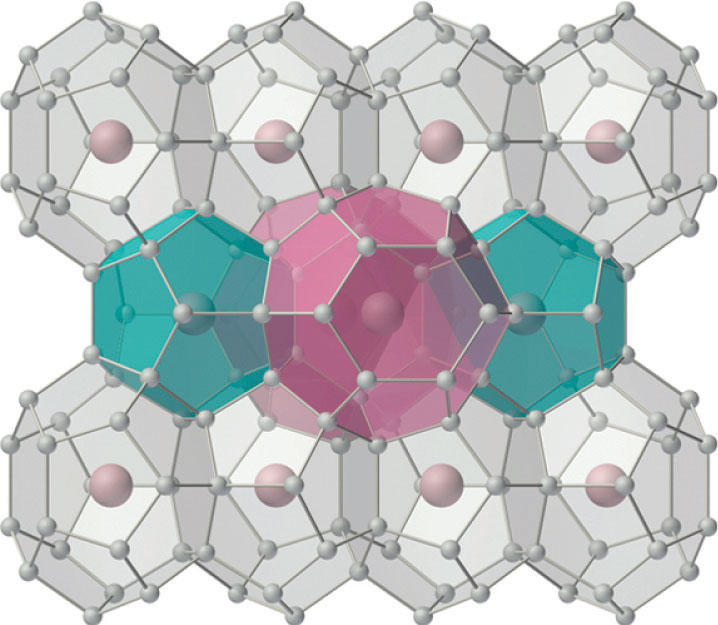
Because clathrate formation does not involve the formation of chemical bonds between the guest (Xe) and the host molecules (H2O, in the case of xenon hydrate), the guest molecules are immediately released when the clathrate is melted or dissolved.
Methane Clathrates
In addition to the noble gases, many other species form stable clathrates. One of the most interesting is methane hydrate, large deposits of which occur naturally at the bottom of the oceans. It is estimated that the amount of methane in such deposits could have a major impact on the world’s energy needs later in this century.

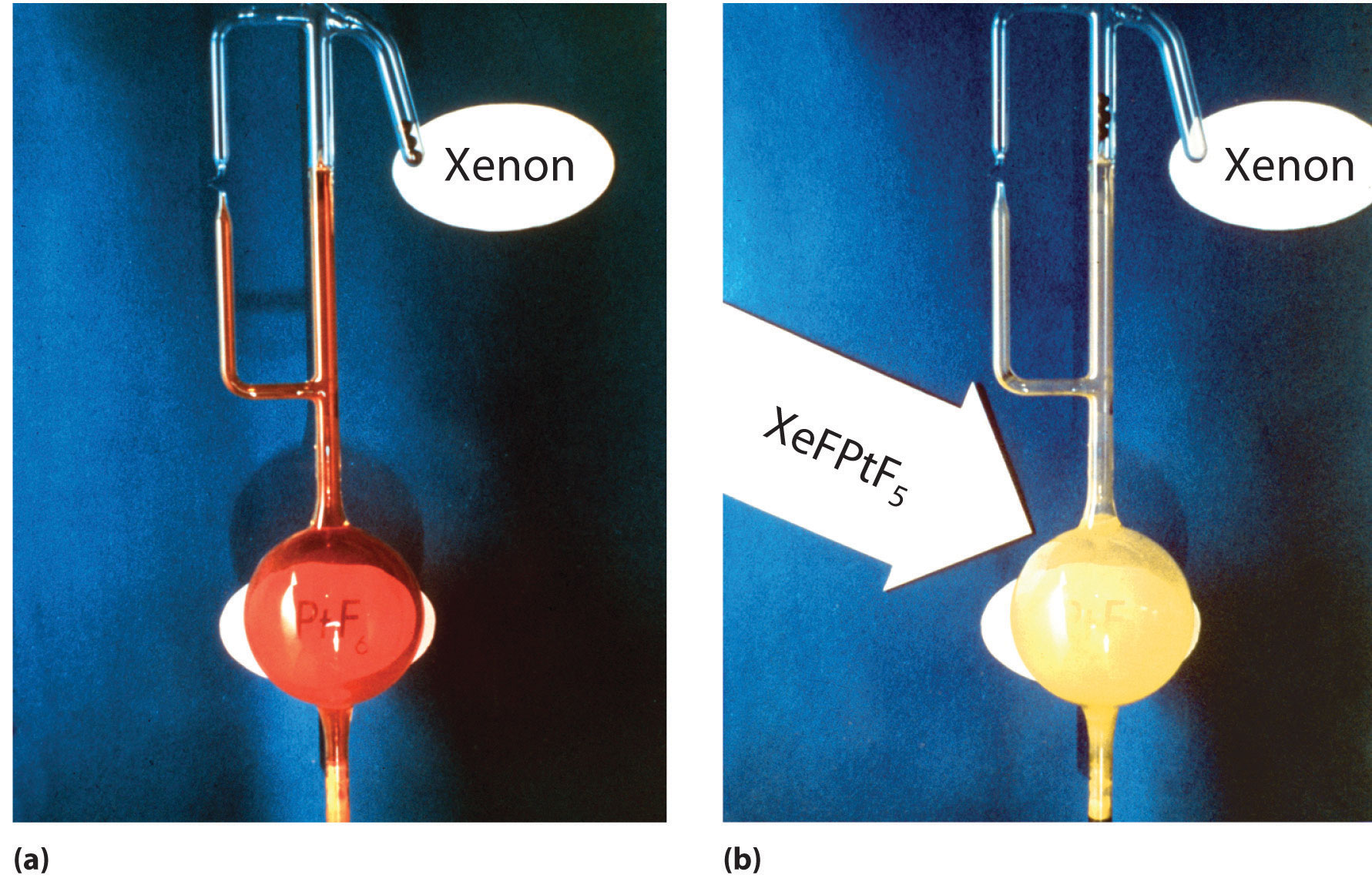
The widely held belief in the intrinsic lack of reactivity of the noble gases was challenged when Neil Bartlett, a British professor of chemistry at the University of British Columbia, showed that PtF6, a compound used in the Manhattan Project, could oxidize O2. Because the ionization energy of xenon (1170 kJ/mol) is actually lower than that of O2, Bartlett recognized that PtF6 should also be able to oxidize xenon. When he mixed colorless xenon gas with deep red PtF6 vapor, yellow-orange crystals immediately formed (Figure \(\PageIndex{3}\)). Although Bartlett initially postulated that they were \(\ce{Xe^{+}PtF6^{−}}\), it is now generally agreed that the reaction also involves the transfer of a fluorine atom to xenon to give the \(\ce{XeF^{+}}\) ion:
\[\ce{Xe(g) + PtF6(g) -> [XeF^{+}][PtF5^{−}](s)} \label{Eq1}\]
Subsequent work showed that xenon reacts directly with fluorine under relatively mild conditions to give XeF2, XeF4, or XeF6, depending on conditions; one such reaction is as follows:
\[\ce{Xe(g) + 2F2(g) -> XeF4(s)} \label{Eq2}\]
The ionization energies of helium, neon, and argon are so high (Table \(\PageIndex{1}\)) that no stable compounds of these elements are known. The ionization energies of krypton and xenon are lower but still very high; consequently only highly electronegative elements (F, O, and Cl) can form stable compounds with xenon and krypton without being oxidized themselves. Xenon reacts directly with only two elements: F2 and Cl2. Although \(\ce{XeCl2}\) and \(\ce{KrF2}\) can be prepared directly from the elements, they are substantially less stable than the xenon fluorides.
The ionization energies of helium, neon, and argon are so high that no stable compounds of these elements are known.
Because halides of the noble gases are powerful oxidants and fluorinating agents, they decompose rapidly after contact with trace amounts of water, and they react violently with organic compounds or other reductants. The xenon fluorides are also Lewis acids; they react with the fluoride ion, the only Lewis base that is not oxidized immediately on contact, to form anionic complexes. For example, reacting cesium fluoride with XeF6 produces CsXeF7, which gives Cs2XeF8 when heated:
\[\ce{XeF6(s) + CsF(s) -> CsXeF7(s)} \label{Eq3}\]
\[\ce{2CsXeF7(s) ->[\Delta] Cs2XeF8(s) + XeF6(g)} \label{Eq4}\]
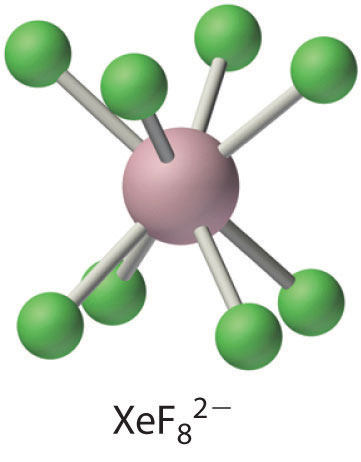
The \(\ce{XeF8^{2-}}\) ion contains eight-coordinate xenon and has the square antiprismatic structure, which is essentially identical to that of the IF8− ion. Cs2XeF8 is surprisingly stable for a polyatomic ion that contains xenon in the +6 oxidation state, decomposing only at temperatures greater than 300°C. Major factors in the stability of Cs2XeF8 are almost certainly the formation of a stable ionic lattice and the high coordination number of xenon, which protects the central atom from attack by other species. (Recall from that this latter effect is responsible for the extreme stability of SF6.)
For a previously “inert” gas, xenon has a surprisingly high affinity for oxygen, presumably because of π bonding between \(O\) and \(Xe\). Consequently, xenon forms an extensive series of oxides and oxoanion salts. For example, hydrolysis of either \(XeF_4\) or \(XeF_6\) produces \(XeO_3\), an explosive white solid:
\[\ce{XeF6(aq) + 3H2O(l) -> XeO3(aq) + 6HF(aq)} \label{Eq5}\]
Treating a solution of XeO3 with ozone, a strong oxidant, results in further oxidation of xenon to give either XeO4, a colorless, explosive gas, or the surprisingly stable perxenate ion (XeO64−), both of which contain xenon in its highest possible oxidation state (+8). The chemistry of the xenon halides and oxides is best understood by analogy to the corresponding compounds of iodine. For example, XeO3 is isoelectronic with the iodate ion (IO3−), and XeF82− is isoelectronic with the IF8− ion.
Xenon has a high affinity for both fluorine and oxygen.
Because the ionization energy of radon is less than that of xenon, in principle radon should be able to form an even greater variety of chemical compounds than xenon. Unfortunately, however, radon is so radioactive that its chemistry has not been extensively explored.
Example \(\PageIndex{1}\)
On a virtual planet similar to Earth, at least one isotope of radon is not radioactive. A scientist explored its chemistry and presented her major conclusions in a trailblazing paper on radon compounds, focusing on the kinds of compounds formed and their stoichiometries. Based on periodic trends, how did she summarize the chemistry of radon?
Given: nonradioactive isotope of radon
Asked for: summary of its chemistry
Strategy:
Based on the position of radon in the periodic table and periodic trends in atomic properties, thermodynamics, and kinetics, predict the most likely reactions and compounds of radon.
Solution
We expect radon to be significantly easier to oxidize than xenon. Based on its position in the periodic table, however, we also expect its bonds to other atoms to be weaker than those formed by xenon. Radon should be more difficult to oxidize to its highest possible oxidation state (+8) than xenon because of the inert-pair effect. Consequently, radon should form an extensive series of fluorides, including RnF2, RnF4, RnF6, and possibly RnF8 (due to its large radius). The ion RnF82− should also exist. We expect radon to form a series of oxides similar to those of xenon, including RnO3 and possibly RnO4. The biggest surprise in radon chemistry is likely to be the existence of stable chlorides, such as RnCl2 and possibly even RnCl4.
Exercise \(\PageIndex{1}\)
Predict the stoichiometry of the product formed by reacting XeF6 with a 1:1 stoichiometric amount of KF and propose a reasonable structure for the anion.
Answer\(\ce{KXeF7}\); the xenon atom in XeF7− has 16 valence electrons, which according to the valence-shell electron-pair repulsion model could give either a square antiprismatic structure with one fluorine atom missing or a pentagonal bipyramid if the 5s2 electrons behave like an inert pair that does not participate in bonding.
Tag » A Reactive Element With A Relatively High Electronegativity
-
Solved QUESTION 10 A Reactive Element With A Relatively High
-
A Reactive Element With A Relatively High ...
-
Chemistry Chapter 7 Review Flashcards | Quizlet
-
A Reactive Element With A Relatively High Electronegativity Would Be ...
-
QUESTION 10 A Reactive Element With A Relatively High … - Study QA
-
2.12: Electronegativity - Chemistry LibreTexts
-
A Reactive Element With A Relatively High Electronegativity Would ...
-
Which Of The Following Elements Have Relatively High ... - Toppr
-
[PDF] 4/4/2015 CHEM 177- Chapter 7 Review TINA AKINYI
-
A Al 2 O 3 B MgO C NaCl D AlF 3 2 8 A Reactive Element With A ...
-
Electronegativity Seen As The Ground-State Average Valence ...
-
Periodic Table - Model Science Software