What Is An Electric Cell Class 11 Physics CBSE - Vedantu
Maybe your like
CoursesCourses for KidsFree study materialOffline CentresMore Store
Store

 Answer
Answer Question Answers for Class 12
Question Answers for Class 12 Class 12 BiologyClass 12 ChemistryClass 12 EnglishClass 12 MathsClass 12 PhysicsClass 12 Social ScienceClass 12 Business StudiesClass 12 EconomicsQuestion Answers for Class 11
Class 12 BiologyClass 12 ChemistryClass 12 EnglishClass 12 MathsClass 12 PhysicsClass 12 Social ScienceClass 12 Business StudiesClass 12 EconomicsQuestion Answers for Class 11 Class 11 EconomicsClass 11 Computer ScienceClass 11 BiologyClass 11 ChemistryClass 11 EnglishClass 11 MathsClass 11 PhysicsClass 11 Social ScienceClass 11 AccountancyClass 11 Business StudiesQuestion Answers for Class 10
Class 11 EconomicsClass 11 Computer ScienceClass 11 BiologyClass 11 ChemistryClass 11 EnglishClass 11 MathsClass 11 PhysicsClass 11 Social ScienceClass 11 AccountancyClass 11 Business StudiesQuestion Answers for Class 10 Class 10 ScienceClass 10 EnglishClass 10 MathsClass 10 Social ScienceClass 10 General KnowledgeQuestion Answers for Class 9
Class 10 ScienceClass 10 EnglishClass 10 MathsClass 10 Social ScienceClass 10 General KnowledgeQuestion Answers for Class 9 Class 9 General KnowledgeClass 9 ScienceClass 9 EnglishClass 9 MathsClass 9 Social ScienceQuestion Answers for Class 8
Class 9 General KnowledgeClass 9 ScienceClass 9 EnglishClass 9 MathsClass 9 Social ScienceQuestion Answers for Class 8 Class 8 ScienceClass 8 EnglishClass 8 MathsClass 8 Social ScienceQuestion Answers for Class 7
Class 8 ScienceClass 8 EnglishClass 8 MathsClass 8 Social ScienceQuestion Answers for Class 7 Class 7 ScienceClass 7 EnglishClass 7 MathsClass 7 Social ScienceQuestion Answers for Class 6
Class 7 ScienceClass 7 EnglishClass 7 MathsClass 7 Social ScienceQuestion Answers for Class 6 Class 6 ScienceClass 6 EnglishClass 6 MathsClass 6 Social ScienceQuestion Answers for Class 5
Class 6 ScienceClass 6 EnglishClass 6 MathsClass 6 Social ScienceQuestion Answers for Class 5 Class 5 ScienceClass 5 EnglishClass 5 MathsClass 5 Social ScienceQuestion Answers for Class 4
Class 5 ScienceClass 5 EnglishClass 5 MathsClass 5 Social ScienceQuestion Answers for Class 4 Class 4 ScienceClass 4 EnglishClass 4 Maths
Class 4 ScienceClass 4 EnglishClass 4 Maths
 What is an electric cell?Answer
What is an electric cell?Answer Verified540.6k+ viewsHint: An electric cell also known as an electrochemical cell is a device that converts chemical energy into electrical energy to do useful work like power a load (i.e. bulbs, etc). Current flows from the positive terminal (cathode) via the external circuit to the negative terminal (anode).Complete answer:Electric cell is an electrical power supply to power the load. There are three types of cell referred to as an electrical cell, an electrochemical cell, a solar cell and an electrolytic cell. The process involved in each is similar and all are mainly used to produce current in the circuit and power the load. The figure below shows a typical electric cell, it has two metal electrodes immersed in an electrolyte solution.
Verified540.6k+ viewsHint: An electric cell also known as an electrochemical cell is a device that converts chemical energy into electrical energy to do useful work like power a load (i.e. bulbs, etc). Current flows from the positive terminal (cathode) via the external circuit to the negative terminal (anode).Complete answer:Electric cell is an electrical power supply to power the load. There are three types of cell referred to as an electrical cell, an electrochemical cell, a solar cell and an electrolytic cell. The process involved in each is similar and all are mainly used to produce current in the circuit and power the load. The figure below shows a typical electric cell, it has two metal electrodes immersed in an electrolyte solution. 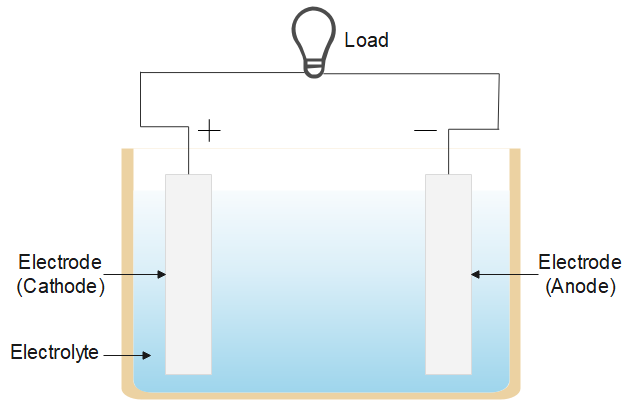 It converts chemical energy into electrical energy, the electrolyte (like, sodium chloride) is a solution of water and solvent containing dissolved ions. The ions get attracted to the electrode of opposite charge and chemical reactions (redox reactions) take place and an electric current is drawn in the circuit.The maximum potential difference between the two electrodes is called electromotive force or the emf of the cell, it is the energy per unit charge. Additional Information: Galvanic cells are a type of electrochemical cell. It consists of two electrodes made of two different metals immersed in separate electrolyte solutions and a salt bridge to connect these two half cells. Note: Electric cells can be rechargeable or use and throw, non rechargeable electric cells are often referred to as primary cells (chemical reactions here are not reversible) and rechargeable electric cells are often referred to as secondary cells.Recently Updated PagesWhy are manures considered better than fertilizers class 11 biology CBSE
It converts chemical energy into electrical energy, the electrolyte (like, sodium chloride) is a solution of water and solvent containing dissolved ions. The ions get attracted to the electrode of opposite charge and chemical reactions (redox reactions) take place and an electric current is drawn in the circuit.The maximum potential difference between the two electrodes is called electromotive force or the emf of the cell, it is the energy per unit charge. Additional Information: Galvanic cells are a type of electrochemical cell. It consists of two electrodes made of two different metals immersed in separate electrolyte solutions and a salt bridge to connect these two half cells. Note: Electric cells can be rechargeable or use and throw, non rechargeable electric cells are often referred to as primary cells (chemical reactions here are not reversible) and rechargeable electric cells are often referred to as secondary cells.Recently Updated PagesWhy are manures considered better than fertilizers class 11 biology CBSE Find the coordinates of the midpoint of the line segment class 11 maths CBSE
Find the coordinates of the midpoint of the line segment class 11 maths CBSE Distinguish between static friction limiting friction class 11 physics CBSE
Distinguish between static friction limiting friction class 11 physics CBSE The Chairman of the constituent Assembly was A Jawaharlal class 11 social science CBSE
The Chairman of the constituent Assembly was A Jawaharlal class 11 social science CBSE The first National Commission on Labour NCL submitted class 11 social science CBSE
The first National Commission on Labour NCL submitted class 11 social science CBSE Number of all subshell of n + l 7 is A 4 B 5 C 6 D class 11 chemistry CBSE
Number of all subshell of n + l 7 is A 4 B 5 C 6 D class 11 chemistry CBSE Why are manures considered better than fertilizers class 11 biology CBSE
Why are manures considered better than fertilizers class 11 biology CBSE Find the coordinates of the midpoint of the line segment class 11 maths CBSE
Find the coordinates of the midpoint of the line segment class 11 maths CBSE Distinguish between static friction limiting friction class 11 physics CBSE
Distinguish between static friction limiting friction class 11 physics CBSE The Chairman of the constituent Assembly was A Jawaharlal class 11 social science CBSE
The Chairman of the constituent Assembly was A Jawaharlal class 11 social science CBSE The first National Commission on Labour NCL submitted class 11 social science CBSE
The first National Commission on Labour NCL submitted class 11 social science CBSE Number of all subshell of n + l 7 is A 4 B 5 C 6 D class 11 chemistry CBSE
Number of all subshell of n + l 7 is A 4 B 5 C 6 D class 11 chemistry CBSE
 One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE Difference Between Prokaryotic Cells and Eukaryotic Cells
Difference Between Prokaryotic Cells and Eukaryotic Cells 1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE
1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE State the laws of reflection of light
State the laws of reflection of light Explain zero factorial class 11 maths CBSE
Explain zero factorial class 11 maths CBSE 10 examples of friction in our daily life
10 examples of friction in our daily life One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE Difference Between Prokaryotic Cells and Eukaryotic Cells
Difference Between Prokaryotic Cells and Eukaryotic Cells 1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE
1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE State the laws of reflection of light
State the laws of reflection of light Explain zero factorial class 11 maths CBSE
Explain zero factorial class 11 maths CBSE
Talk to our experts
1800-120-456-456
Sign In- Question Answer
- Class 11
- Physics
- What is an electric cell


 Question Answers for Class 12
Question Answers for Class 12
 What is an electric cell?Answer
What is an electric cell?Answer It converts chemical energy into electrical energy, the electrolyte (like, sodium chloride) is a solution of water and solvent containing dissolved ions. The ions get attracted to the electrode of opposite charge and chemical reactions (redox reactions) take place and an electric current is drawn in the circuit.The maximum potential difference between the two electrodes is called electromotive force or the emf of the cell, it is the energy per unit charge. Additional Information: Galvanic cells are a type of electrochemical cell. It consists of two electrodes made of two different metals immersed in separate electrolyte solutions and a salt bridge to connect these two half cells. Note: Electric cells can be rechargeable or use and throw, non rechargeable electric cells are often referred to as primary cells (chemical reactions here are not reversible) and rechargeable electric cells are often referred to as secondary cells.Recently Updated PagesWhy are manures considered better than fertilizers class 11 biology CBSE
It converts chemical energy into electrical energy, the electrolyte (like, sodium chloride) is a solution of water and solvent containing dissolved ions. The ions get attracted to the electrode of opposite charge and chemical reactions (redox reactions) take place and an electric current is drawn in the circuit.The maximum potential difference between the two electrodes is called electromotive force or the emf of the cell, it is the energy per unit charge. Additional Information: Galvanic cells are a type of electrochemical cell. It consists of two electrodes made of two different metals immersed in separate electrolyte solutions and a salt bridge to connect these two half cells. Note: Electric cells can be rechargeable or use and throw, non rechargeable electric cells are often referred to as primary cells (chemical reactions here are not reversible) and rechargeable electric cells are often referred to as secondary cells.Recently Updated PagesWhy are manures considered better than fertilizers class 11 biology CBSE Find the coordinates of the midpoint of the line segment class 11 maths CBSE
Find the coordinates of the midpoint of the line segment class 11 maths CBSE Distinguish between static friction limiting friction class 11 physics CBSE
Distinguish between static friction limiting friction class 11 physics CBSE The Chairman of the constituent Assembly was A Jawaharlal class 11 social science CBSE
The Chairman of the constituent Assembly was A Jawaharlal class 11 social science CBSE The first National Commission on Labour NCL submitted class 11 social science CBSE
The first National Commission on Labour NCL submitted class 11 social science CBSE Number of all subshell of n + l 7 is A 4 B 5 C 6 D class 11 chemistry CBSE
Number of all subshell of n + l 7 is A 4 B 5 C 6 D class 11 chemistry CBSE Why are manures considered better than fertilizers class 11 biology CBSE
Why are manures considered better than fertilizers class 11 biology CBSE Find the coordinates of the midpoint of the line segment class 11 maths CBSE
Find the coordinates of the midpoint of the line segment class 11 maths CBSE Distinguish between static friction limiting friction class 11 physics CBSE
Distinguish between static friction limiting friction class 11 physics CBSE The Chairman of the constituent Assembly was A Jawaharlal class 11 social science CBSE
The Chairman of the constituent Assembly was A Jawaharlal class 11 social science CBSE The first National Commission on Labour NCL submitted class 11 social science CBSE
The first National Commission on Labour NCL submitted class 11 social science CBSE Number of all subshell of n + l 7 is A 4 B 5 C 6 D class 11 chemistry CBSE
Number of all subshell of n + l 7 is A 4 B 5 C 6 D class 11 chemistry CBSE
- 1
- 2
 One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE Difference Between Prokaryotic Cells and Eukaryotic Cells
Difference Between Prokaryotic Cells and Eukaryotic Cells 1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE
1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE State the laws of reflection of light
State the laws of reflection of light Explain zero factorial class 11 maths CBSE
Explain zero factorial class 11 maths CBSE 10 examples of friction in our daily life
10 examples of friction in our daily life One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE Difference Between Prokaryotic Cells and Eukaryotic Cells
Difference Between Prokaryotic Cells and Eukaryotic Cells 1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE
1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE State the laws of reflection of light
State the laws of reflection of light Explain zero factorial class 11 maths CBSE
Explain zero factorial class 11 maths CBSE
- 1
- 2
Repeaters Course for NEET 2022 - 23
NEET Repeater 2023 - Aakrosh 1 Year CourseTag » How Does An Electric Cell Work
-
Electrical Cell - Isaac Physics
-
What Is An Electric Cell: Working And Example - EMBIBE
-
Simple Electrical Cell - MagLab
-
O-Level Chemistry: Simple Electric Cells
-
How A Battery Works - Curious - Australian Academy Of Science
-
Electric Cell | Physics - YouTube
-
Electrical Cell - Simple English Wikipedia, The Free Encyclopedia
-
What Is An Electric Cell? - Quora
-
16.1 Electric Cells | Electric Cells As Energy Systems - Siyavula
-
How Do Batteries Work? A Simple Introduction - Explain That Stuff
-
How Do Batteries Work?
-
What Is An Electric Cell? Q&A - Byju's
-
The Electric Cell Is A Device To Obtain A Electric Class 12 Physics CBSE
-
How The Human Body Uses Electricity