Enzyme-catalyzed Protein Crosslinking - PubMed
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
Access keys NCBI Homepage MyNCBI Homepage Main Content Main Navigation Search: Search Advanced Clipboard Save Email Send to
Search: Search Advanced Clipboard Save Email Send to- Clipboard
- My Bibliography
- Collections
- Citation manager
Save citation to file
Format: Summary (text) PubMed PMID Abstract (text) CSV Create file CancelEmail citation
Email address has not been verified. Go to My NCBI account settings to confirm your email and then refresh this page. To: Subject: Body: Format: Summary Summary (text) Abstract Abstract (text) MeSH and other data Send email CancelAdd to Collections
- Create a new collection
- Add to an existing collection
Add to My Bibliography
- My Bibliography
Your saved search
Name of saved search: Search terms: Test search terms Would you like email updates of new search results? Saved Search Alert Radio Buttons- Yes
- No
Create a file for external citation management software
Create file CancelYour RSS Feed
Name of RSS Feed: Number of items displayed: 5 10 15 20 50 100 Create RSS Cancel RSS Link CopyFull text links
 Springer Free PMC article Full text links
Springer Free PMC article Full text links Actions
CiteCollectionsAdd to Collections- Create a new collection
- Add to an existing collection
Page navigation
- Title & authors
- Abstract
- Figures
- References
- Publication types
- MeSH terms
- Substances
- LinkOut - more resources
Abstract
The process of protein crosslinking comprises the chemical, enzymatic, or chemoenzymatic formation of new covalent bonds between polypeptides. This allows (1) the site-directed coupling of proteins with distinct properties and (2) the de novo assembly of polymeric protein networks. Transferases, hydrolases, and oxidoreductases can be employed as catalysts for the synthesis of crosslinked proteins, thereby complementing chemical crosslinking strategies. Here, we review enzymatic approaches that are used for protein crosslinking at the industrial level or have shown promising potential in investigations on the lab-scale. We illustrate the underlying mechanisms of crosslink formation and point out the roles of the enzymes in their natural environments. Additionally, we discuss advantages and drawbacks of the enzyme-based crosslinking strategies and their potential for different applications.
PubMed Disclaimer
Figures

Fig. 1
Schematic illustration of enzyme-catalyzed covalent…
Fig. 1
Schematic illustration of enzyme-catalyzed covalent modifications of proteins in vivo; for a more…

Fig. 2
Simplified schematic illustration of the…
Fig. 2
Simplified schematic illustration of the enzymatic cascade leading to ubiquitination of target proteins…
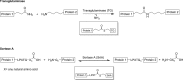
Fig. 3
Protein crosslinking through transamidation reactions…
Fig. 3
Protein crosslinking through transamidation reactions catalyzed by transglutaminase and sortase A. The reactive…

Fig. 4
Crosslinking of proteins mediated through…
Fig. 4
Crosslinking of proteins mediated through oxidation by oxidoreductases. The reactive species generated by…
References
-
- Ando H, Adachi M, Umeda K, Matsuura A, Nonaka M, Uchio R, Tanaka H, Motoki M. Purification and characteristics of a novel transglutaminase derived from microorganisms. Agr Biol Chem Tokyo. 1989;53:2613–2617. doi: 10.1271/bbb1961.53.2613. - DOI - PMC - PubMed
-
- Autio K, Kruus K, Knaapila A, Gerber N, Flander L, Buchert J. Kinetics of transglutaminase-induced cross-linking of wheat proteins in dough. J Agric Food Chem. 2005;53:1039–1045. doi: 10.1021/jf0485032. - DOI - PubMed
-
- Basman A, Köksel H, Ng PKW. Effects of transglutaminase on SDS-PAGE patterns of wheat, soy, and barley proteins and their blends. J Food Sci. 2002;67:2654–2658. doi: 10.1111/j.1365-2621.2002.tb08794.x. -
Từ khóa » đạm Enzyme
-
Protein And Enzyme Activity Assays | Thermo Fisher Scientific - SG
-
Action Of Multi-enzyme Complex On Protein Extraction To Obtain A ...
-
UbiCREST Enzyme Set Protein K-400 - R&D Systems
-
Enzyme And Protein - Home - Karger Publishers
-
Gel Plus Enzyme Protein Gel 2 Oz. - Epicuren Discovery
-
Enzyme Assisted Protein Extraction From Rapeseed, Soybean, And ...
-
AirID, A Novel Proximity Biotinylation Enzyme, For Analysis Of Protein ...
-
Protein, Enzyme, And ELISA Kits | Bio-Rad
-
Protein, Enzyme, And ELISA Kits - Bio-Rad
-
Enzyme Encapsulation By Protein Cages - RSC Publishing
-
Highly Efficient Enzyme Encapsulation In A Protein Nanocage
-
Protein AND Enzyme Gated Supramolecular Disassembly
-
Enzyme-catalyzed Protein Crosslinking | SpringerLink